Research Articles
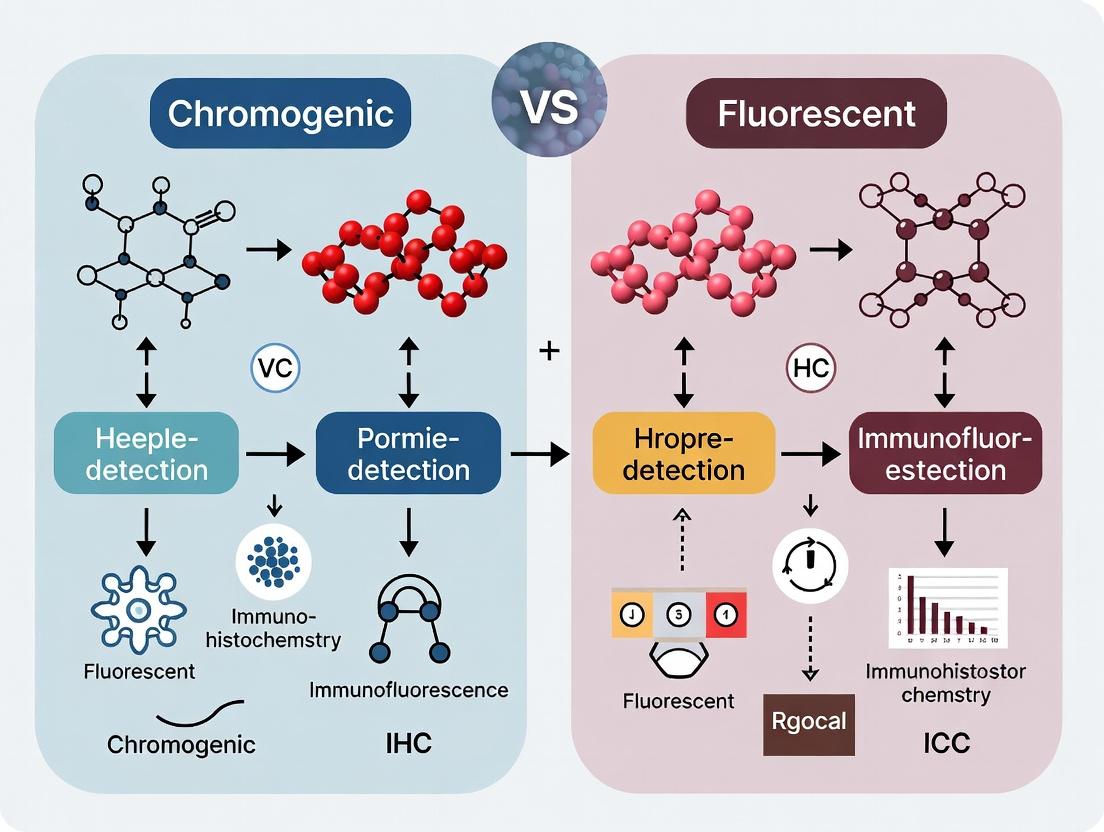
Chromogenic vs. Fluorescent IHC/ICC: A Complete Guide for Researchers in Biomarker Detection
This article provides a comprehensive comparison of chromogenic (DAB) and fluorescent detection methods in immunohistochemistry (IHC) and immunocytochemistry (ICC).
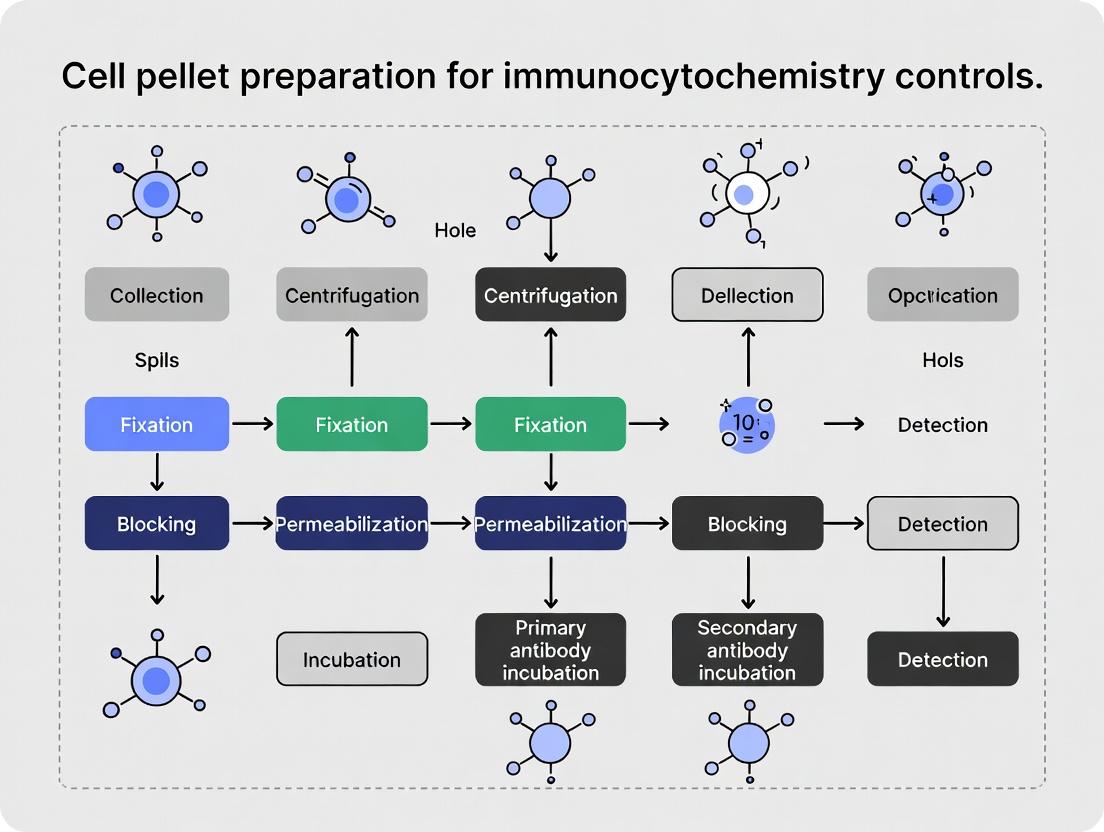
Immunocytochemistry Control Pellet Guide: Essential Protocol for Validation in Research & Drug Development
This comprehensive guide details the critical role of cell pellet preparation in generating robust controls for immunocytochemistry (ICC).
A Practical Guide to CLSI IHC Validation for CLIA Compliance: Protocols, Optimization, and Best Practices
This comprehensive guide details the application of Clinical and Laboratory Standards Institute (CLSI) guidelines for the validation and verification of immunohistochemistry (IHC) assays within Clinical Laboratory Improvement Amendments (CLIA)-certified laboratories.
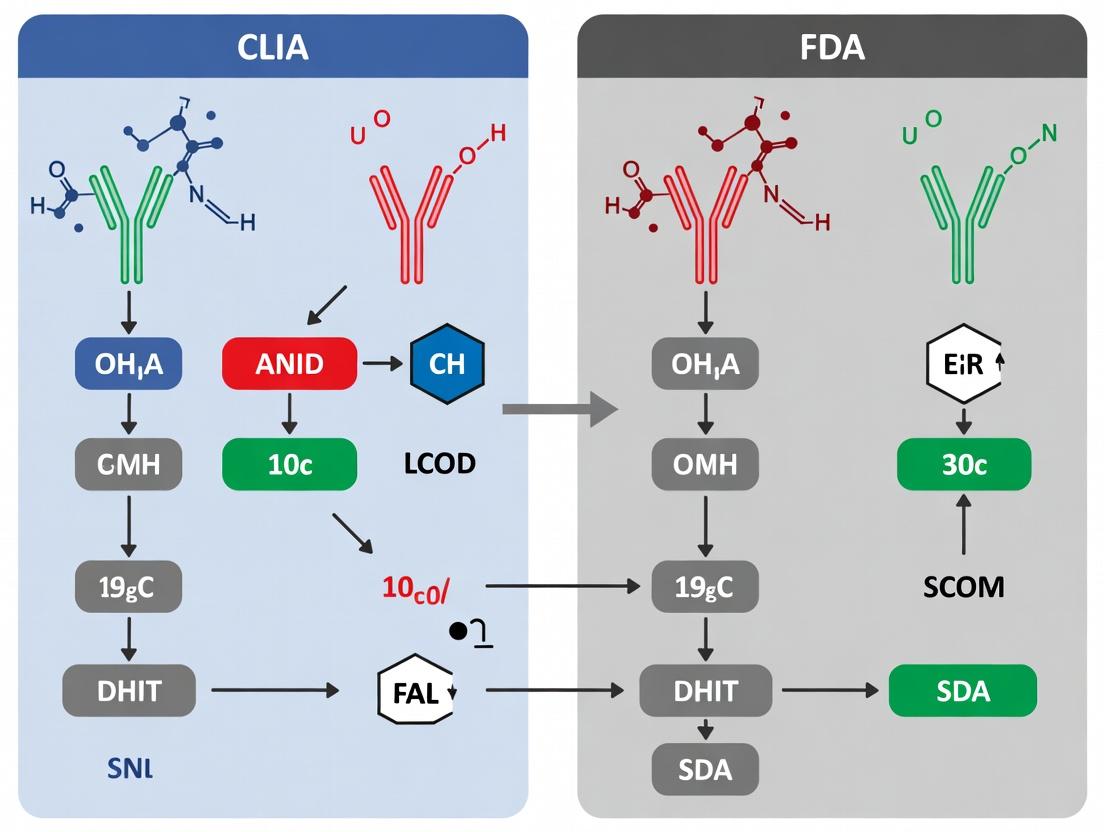
CLIA vs FDA for IHC Assays: A Definitive Guide for Research and Diagnostic Compliance
This article provides a comprehensive analysis of CLIA and FDA regulatory requirements for immunohistochemistry (IHC) assays, tailored for researchers, scientists, and drug development professionals.
Navigating the Regulatory Shift: A Comprehensive Guide to CLIA Validation vs. IVDR for IHC Assays in Research and Drug Development
This article provides a critical comparison between the Clinical Laboratory Improvement Amendments (CLIA) validation framework, predominant in the US, and the In Vitro Diagnostic Regulation (IVDR), the new EU regulatory...
A Practical Guide to CLIA Validation for IHC Assays: From Design to Compliance
This article provides a comprehensive, step-by-step framework for designing and executing a robust CLIA validation study for immunohistochemistry (IHC) assays.
Navigating CLIA Validation for IHC Predictive Biomarkers: A Comprehensive Guide for Precision Medicine
This article provides a detailed roadmap for researchers and drug development professionals to successfully validate immunohistochemistry (IHC) predictive markers under Clinical Laboratory Improvement Amendments (CLIA) requirements.
Navigating CLIA Validation for IHC Assays: A Complete Guide for Research and Development
This comprehensive guide demystifies the process of achieving CLIA (Clinical Laboratory Improvement Amendments) validation for Immunohistochemistry (IHC) assays.
Beyond Formalin: A Practical Guide to CLIA Validation for IHC Assays on Alternative Fixatives
This comprehensive guide addresses the critical need for validating immunohistochemistry (IHC) assays on alternative tissue fixatives under CLIA (Clinical Laboratory Improvement Amendments) regulations.
Navigating CLIA Compliance: A Complete Guide to IHC Assay Revalidation for Clinical Labs
This comprehensive guide demystifies CLIA regulations for Immunohistochemistry (IHC) assay revalidation.