Achieving Multi-Site Reproducibility in IHC: A Complete Guide to Robust Assay Development
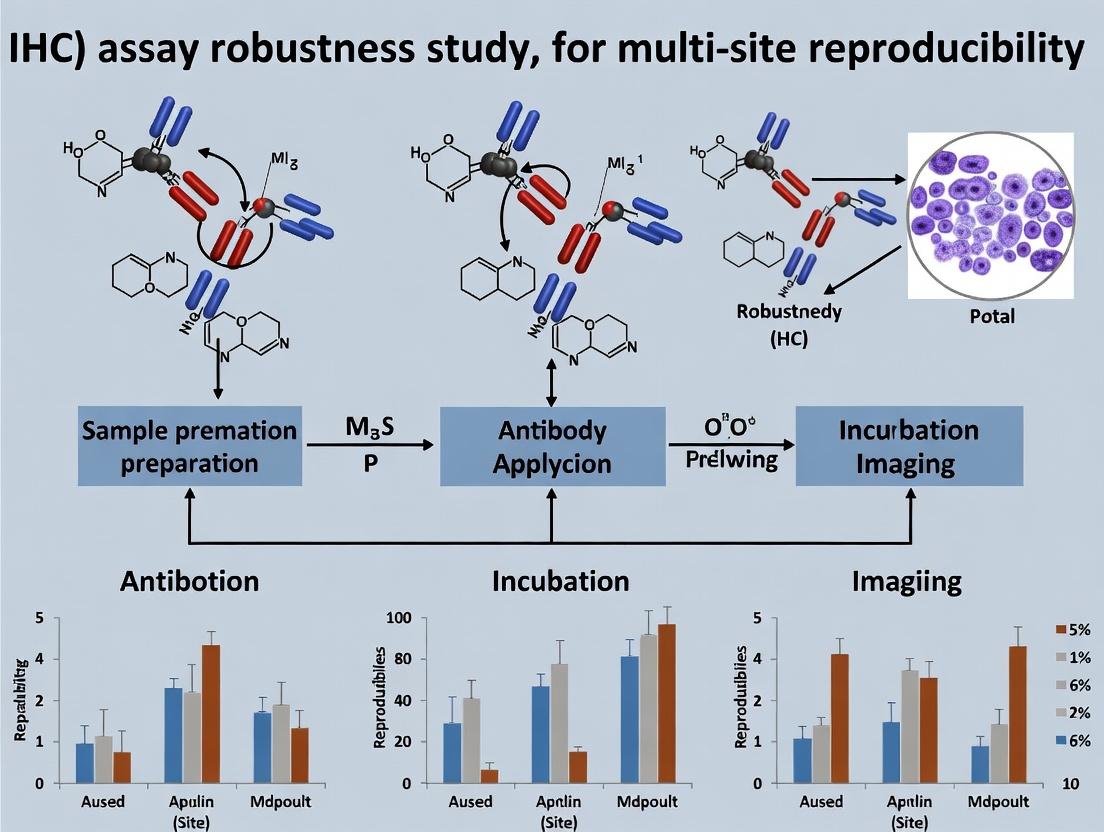
This comprehensive guide addresses the critical challenge of ensuring immunohistochemistry (IHC) assay robustness and reproducibility across multiple laboratory sites in research and drug development.
Achieving Multi-Site Reproducibility in IHC: A Complete Guide to Robust Assay Development
Abstract
This comprehensive guide addresses the critical challenge of ensuring immunohistochemistry (IHC) assay robustness and reproducibility across multiple laboratory sites in research and drug development. We explore the foundational importance of reproducibility in translational science, detail actionable methodological frameworks for standardizing pre-analytical, analytical, and post-analytical variables, provide systematic troubleshooting strategies for common cross-site discrepancies, and review validation protocols and comparative analyses of standardization tools. Designed for researchers, scientists, and drug development professionals, this article synthesizes current best practices and emerging standards to enable reliable, comparable IHC data in multi-center studies.
Why Reproducibility Fails: The Foundational Challenge of Multi-Site IHC
Defining Robustness and Reproducibility in the IHC Context
Technical Support Center: Troubleshooting Guides and FAQs
Frequently Asked Questions
Q1: What are the most critical pre-analytical variables affecting IHC robustness across multiple sites? A: Pre-analytical variability is the primary source of non-reproducibility. Key variables include:
- Tissue Fixation: Delay, duration, and type of fixative (e.g., 10% NBF) must be standardized.
- Tissue Processing: Time in dehydrating alcohols and clearing agents.
- Embedding: Orientation and paraffin block temperature.
- Sectioning: Section thickness (typically 4-5 µm) and water bath temperature.
- Antigen Retrieval: Method (heat-induced vs. enzymatic), pH of buffer, and retrieval time.
Q2: How can we minimize inter-observer scoring variability in a multi-site study? A: Implement a standardized scoring protocol:
- Use validated, continuous digital image analysis (DIA) where possible.
- If manual scoring is required, employ a semi-quantitative method (e.g., H-score) with defined thresholds.
- Require all scorers from all sites to undergo centralized, rigorous training using a shared set of reference slides.
- Perform regular concordance testing (e.g., Cohen's kappa) between scorers and sites.
Q3: Our positive controls are inconsistent. What steps should we take? A: Inconsistent controls indicate a lack of assay robustness. Troubleshoot using this hierarchy:
- Verify reagent integrity: Check antibody and detection kit expiry dates. Use fresh, aliquoted buffers.
- Check equipment calibration: Validate oven/water bath/steamer temperature and pressure cooker pressure.
- Optimize antibody titration: Re-titrate the primary antibody on a known positive control tissue using a checkerboard dilution series.
- Standardize the detection system: Ensure consistent incubation times and temperatures for all detection steps.
Q4: What is the best way to validate an antibody for a multi-site reproducibility study? A: Follow a orthogonal validation framework:
- Confirm specificity: Use siRNA/CRISPR knockout cell lines or genetically modified tissue as a negative control.
- Verify expected localization: Compare staining pattern to published data and molecular databases.
- Assay precision: Perform intra-run, inter-run, inter-operator, and inter-site precision studies.
- Correlate with another method: Where possible, correlate IHC results with an orthogonal method (e.g., mRNA in situ hybridization, Western blot on lysates from the same tissue type).
Troubleshooting Guide: Common IHC Staining Issues
| Problem | Possible Causes | Recommended Action |
|---|---|---|
| Weak or No Staining | Depleted antibody, incorrect retrieval, inactive detection system, over-fixation. | Run a multi-tissue control block. Increase primary antibody concentration; optimize retrieval pH/time. Test detection system with a known robust antibody. |
| High Background | Non-specific binding, over-concentrated antibody, inadequate blocking, drying of sections. | Titrate primary antibody. Extend serum/protein block step. Ensure sections are never allowed to dry post-retrieval. Include relevant isotype control. |
| Non-Specific Nuclear Staining | Often due to over-retrieval or endogenous biotin (in older systems). | Reduce retrieval time. Use a polymer-based, biotin-free detection system. |
| Patchy/Uneven Staining | Inconsistent contact during incubation, bubbles under coverslip, uneven heating during retrieval. | Use automated staining if available. Ensure coverslips are properly applied. Check water bath/steamer for hot spots. |
| Inter-Site Result Discrepancy | Uncalibrated equipment, different reagent lots, subjective scoring. | Implement a Site Qualification Protocol (see below). Centralize critical reagents. Use digital image analysis with a shared algorithm. |
Table 1: Impact of Pre-Analytical Variables on IHC Staining Intensity (H-Score)
| Variable | Standardized Protocol | Non-Standardized Range | % Coefficient of Variation (CV) |
|---|---|---|---|
| Fixation Time (10% NBF) | 18-24 hours | 6 hours - 72 hours | 35-45% |
| Section Thickness | 4 µm | 3 - 6 µm | 22% |
| Antigen Retrieval pH | pH 6.0 | pH 6.0 - pH 9.0 | 50-60% |
| Primary Antibody Incubation | 32 min (automated) | 30 min - overnight (manual) | 25% |
Table 2: Inter-Site Reproducibility Metrics for a Validated PD-L1 IHC Assay
| Performance Metric | Target | Observed Result (n=5 sites) |
|---|---|---|
| Inter-Site Concordance (Positive vs. Negative) | >90% | 98% |
| Intra-Site Precision (CV of H-Score) | <15% | 8% |
| Inter-Site Precision (CV of H-Score) | <20% | 12% |
| Inter-Observer Scoring Agreement (Kappa) | >0.80 | 0.89 |
Experimental Protocols
Protocol 1: Site Qualification for Multi-Center IHC Studies
Purpose: To ensure all participating laboratories achieve equivalent staining results before study initiation. Materials: Centralized kit of validated reagents (antibody, detection kit, buffers), multi-tissue microarray (TMA) control slide. Method:
- Pre-Study Run: All sites process the same TMA slide using the locked, detailed protocol.
- Digital Upload: Sites digitize slides using a calibrated scanner at 20x magnification.
- Central Analysis: A lead site analyzes all digital images using a pre-defined DIA algorithm.
- Acceptance Criteria: Staining intensity (H-score) for each core must fall within ±15% of the lead site's mean. Positive/negative calls must be 100% concordant.
- Remediation: Sites outside criteria repeat training and the qualification run.
Protocol 2: Checkerboard Antibody Titration for Optimization
Purpose: To empirically determine the optimal primary antibody concentration and retrieval conditions. Materials: Positive control tissue, primary antibody, range of retrieval buffers (pH 6-10). Method:
- Cut serial sections from the control block.
- Perform antigen retrieval using three different buffer pH levels (e.g., 6, 8, 9) on separate sections.
- For each retrieval condition, apply a series of primary antibody dilutions (e.g., 1:50, 1:100, 1:200, 1:500, 1:1000).
- Complete staining with standardized detection and visualization.
- Select the condition (pH + dilution) that yields the strongest specific signal with the lowest background. This is the "peak" dilution for use in the validated assay.
Diagrams
Diagram Title: The Three Pillars of IHC Robustness
Diagram Title: Multi-Site IHC Reproducibility Study Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Importance for Robustness |
|---|---|
| Validated Primary Antibody | The core reagent. Must be specific, sensitive, and tested for IHC on fixed tissue. Use clones with known performance data. |
| Polymer-Based Detection System | Provides high sensitivity and low background. Eliminates endogenous biotin interference common in avidin-biotin systems. |
| pH-Calibrated Antigen Retrieval Buffers | Critical for epitope exposure. Using a consistent, precisely formulated buffer (e.g., citrate pH 6.0, Tris-EDTA pH 9.0) is essential. |
| Multi-Tissue Control Block (MTCB) | Contains cell lines or tissues with known expression levels (negative, weak, moderate, strong). Run on every slide/ batch for run-to-run monitoring. |
| Automated Staining Platform | Dramatically improves reproducibility by standardizing incubation times, temperatures, and wash volumes across runs and operators. |
| Digital Slide Scanner & Analysis Software | Enables objective, quantitative assessment of staining (H-score, % positive cells). Facilitates remote review and inter-site comparison. |
| Isotype Control Antibody | Distinguishes specific signal from non-specific background staining. Crucial for assay development and validation. |
Technical Support Center: Troubleshooting IHC Reproducibility
FAQs & Troubleshooting Guides
Q1: Our multi-site trial shows high inter-site variance in HER2 IHC H-scores for the same patient samples. What are the most likely pre-analytical variables? A: Pre-analytical variables are a primary source of irreproducibility. Key factors include:
- Fixation Delay & Duration: Time from biopsy to fixation and total fixation time in 10% Neutral Buffered Formalin must be standardized.
- Fixation Type: Inconsistent use of NBF vs. other fixatives alters epitope availability.
- Ischemic Time: Warm vs. cold ischemia time affects protein degradation.
- Sample Size: Thickness of tissue impacts formalin penetration.
Table 1: Impact of Pre-Analytical Variables on IHC Results
| Variable | Recommended Standard | Effect of Deviation | Typical Impact on H-Score Variance |
|---|---|---|---|
| Fixation Delay | < 1 hour | Antigen degradation/modification | Increase of 15-25% |
| Fixation Duration | 6-72 hours (NBF) | Under-fixation or over-masking | Increase of 20-40% |
| Tissue Thickness | 3-5 mm | Incomplete fixation center | Increase of 25-35% |
| Ischemic Time | Minimize (< 60 min) | Hypoxia-induced changes | Increase of 10-20% |
Protocol: Standardized Pre-Analytical Processing for Multi-Site Studies
- Biopsy Collection: Use a calibrated punch/needle. Immediately place specimen in pre-labeled container.
- Fixation: Immerse in a 10X volume of pre-validated 10% NBF within 60 minutes of excision.
- Fixation Duration: Maintain fixation at room temperature (18-25°C) for 18-24 hours.
- Processing: Use a validated, automated tissue processor with identical dehydration and paraffin-embedding protocols across all sites.
- Sectioning: Cut sections at 4-5 µm thickness using microtomes with freshly replaced blades. Float sections in a 40°C water bath.
- Slide Storage: Store unstained slides at 4°C in a desiccated environment for ≤ 4 weeks before staining.
Q2: During assay validation, our positive controls stain weakly while negative controls show high background. How do we troubleshoot the staining protocol itself? A: This indicates an issue with antigen retrieval and/or detection system conditions.
Table 2: Troubleshooting Staining Protocol Issues
| Symptom | Possible Cause | Corrective Action |
|---|---|---|
| Weak positive control | Suboptimal antigen retrieval | Optimize retrieval time/pH; validate with a range of retrieval buffers (e.g., citrate pH 6.0, Tris-EDTA pH 9.0). |
| High background | Over-retrieval, excessive primary Ab concentration, or inadequate blocking | Titrate primary antibody; implement protein block (e.g., 5% normal serum, casein); optimize retrieval time. |
| Inconsistent staining | Manual staining variability, reagent depletion | Automate staining steps using a validated platform; establish strict reagent lot tracking and changeover protocols. |
| High inter-slide variance | Uneven heating during retrieval, inconsistent washing | Use a calibrated, water bath-based or pressurized retrieval system; implement automated slide washers. |
Protocol: Optimized Antigen Retrieval & Staining Workflow
- Deparaffinization: Standard xylene and ethanol series.
- Antigen Retrieval: Use a pressure cooker or commercial decloaking chamber with 1X Tris-EDTA, pH 9.0, at 95-100°C for 20 minutes. Cool for 30 minutes at room temperature.
- Peroxidase Block: 3% H₂O₂ for 10 minutes.
- Protein Block: Apply 2.5% normal horse serum for 20 minutes.
- Primary Antibody: Apply validated, pre-diluted monoclonal antibody (e.g., anti-PD-L1, clone 22C3) for 60 minutes at room temperature.
- Detection: Use a polymer-based detection system (e.g., HRP-polymer) for 30 minutes.
- Chromogen: Apply DAB substrate for exactly 5 minutes. Monitor development microscopically.
- Counterstain: Hematoxylin for 30 seconds.
Q3: How do we standardize scoring across multiple pathologists to reduce observer bias? A: Implement digital pathology and AI-assisted quantitation with rigorous training.
Table 3: Methods to Reduce Scoring Variance
| Method | Description | Estimated Reduction in Inter-Observer Variance |
|---|---|---|
| Manual + Training | Use standardized scoring guides (e.g., ASCO/CAP) with joint training sessions. | 20-30% |
| Image Analysis Algorithms | Deploy validated digital algorithms for quantitation of % positivity, H-score, or combined positive score (CPS). | 50-70% |
| Continuous Scoring | Replace categorical scores (0, 1+, 2+, 3+) with continuous metrics (e.g., H-score: 0-300). | 30-40% |
Protocol: Digital Image Analysis for PD-L1 CPS Quantification
- Slide Digitization: Scan slides at 20X magnification using a validated whole slide scanner.
- Region of Interest (ROI) Annotation: A certified pathologist digitally annotates viable tumor areas.
- Algorithm Application: Apply a validated algorithm to:
- Segment tumor cells and associated inflammatory cells.
- Detect membranous (and/or cytoplasmic) DAB signal.
- Calculate CPS = (Number of PD-L1 staining cells (tumor cells, lymphocytes, macrophages) / Total number of viable tumor cells) x 100.
- Review & Sign-off: Pathologist reviews algorithm output and approves the final score.
Signaling Pathway & Experimental Workflow Diagrams
Title: Variables Affecting IHC Reproducibility Across Phases
Title: Workflow for Robust Multi-Site IHC
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for Reproducible IHC Assays
| Item | Function & Importance for Reproducibility |
|---|---|
| Validated Primary Antibody Clone | Specific clone (e.g., HER2 clone 4B5, PD-L1 clone 22C3) is critical. Use same clone, lot, and vendor across sites. Defines assay specificity. |
| Isotype-Matched Control IgG | Negative control to distinguish non-specific background from specific signal. Must match host species and isotype of primary antibody. |
| Polymer-Based Detection System | Amplifies signal with high sensitivity and low background. More reproducible than older avidin-biotin systems. |
| Buffered Antigen Retrieval Solution | Standardizes epitope recovery. Citrate (pH 6.0) or Tris-EDTA (pH 9.0) buffer choice is target-dependent. |
| Automated IHC Stainer | Eliminates manual timing and application inconsistencies. Essential for multi-site trials. |
| Whole Slide Scanner | Digitizes slides for archiving, remote review, and application of digital image analysis algorithms. |
| Image Analysis Software | Provides objective, quantitative metrics (H-score, CPS, % positivity), reducing observer bias. |
| Multitissue Control Block | Contains cell lines/tissues with known expression levels (negative, low, high) for run-to-run validation. |
Troubleshooting Guides & FAQs
Pre-Analytical Phase FAQ
Q1: Our IHC staining intensity varies significantly between sites, even with the same tissue type. What pre-analytical factors should we investigate first? A: The most common pre-analytical culprits are tissue fixation and ischemia time. Standardize cold ischemia time to ≤60 minutes and fix in 10% Neutral Buffered Formalin for 24-48 hours (depending on tissue thickness). Use a fixation timer and log deviations.
Q2: How can we ensure consistent antigen retrieval across multiple laboratories? A: Variability often stems from pH drift of retrieval buffers and heating method inconsistency. Implement a protocol using a pressure cooker or declared steamer at 121°C for 15 minutes. Use a validated, pre-mixed EDTA or citrate buffer (pH 6.0 or 9.0) with a strict shelf-life and pH check before each use.
Analytical Phase FAQ
Q3: Our automated stainers show declining signal over time despite using the same protocol. What is the likely cause? A: This typically indicates reagent degradation or instrument calibration drift. First, check the primary antibody dilution stability (often ≤1 week at 4°C). Then, validate the detection system (polymer-HRP) activity using a control slide. Ensure the stainer's liquid dispense volumes are calibrated quarterly.
Q4: How do we troubleshoot high background or non-specific staining? A: High background often results from inadequate blocking or over-fixation. Implement a two-step block: 3% H2O2 for endogenous peroxidase, followed by 5% normal serum from the species of the secondary antibody for 30 minutes. If the issue persists, titrate the primary antibody concentration using a multi-tiered dilution series.
Post-Analytical Phase FAQ
Q5: Scoring discrepancies between pathologists are affecting our study's reproducibility. How can we mitigate this? A: This is a major post-analytical challenge. Implement a mandatory digital pathology training session using a consensus slide set (≥20 images). Utilize standardized scoring algorithms (e.g., H-score, Allred score) and require a minimum inter-rater reliability score (Cohen's kappa >0.7) before study initiation.
Q6: Our digital image analysis yields different results from the same slide when scanned on different days. A: This indicates variability in whole slide image scanner calibration or analysis settings. Calibrate the scanner's light source and camera monthly. For analysis, lock all software parameters (e.g., color threshold, tissue detection sensitivity) and re-validate using the same control slide for every batch scan.
Table 1: Impact of Pre-Analytical Variables on IHC Signal Integrity
| Variable | Acceptable Range | % Signal Loss Outside Range | Recommended Mitigation |
|---|---|---|---|
| Cold Ischemia Time | ≤ 60 min | 15-40% per hour delay | Use cold transport media, strict SOP timing |
| Fixation Time | 18-48 hrs (depends on tissue) | Up to 60% loss | Implement fixation timer alarms |
| Fixative Type | 10% NBF only | N/A | Centralized procurement of fixative |
| Tissue Processing Temp | Ambient (22-25°C) | Variable | Use monitored, calibrated processors |
Table 2: Analytical Phase Reagent Stability & Performance
| Reagent | Optimal Storage | Max Stable Duration After Prep | Key Performance Check |
|---|---|---|---|
| Primary Antibody (diluted) | 4°C, aliquoted | 7 days | Titration curve every new lot |
| Polymer-Based Detection System | 4°C, original bottle | Until expiry | Positive control slide with each run |
| Chromogen (DAB) | Protect from light, 4°C | 24 hrs after prep | Monitor for precipitate formation |
| Antigen Retrieval Buffer (pH 9.0) | RT, sealed | 1 month | Measure pH before each use (target pH ±0.2) |
Experimental Protocols
Protocol 1: Systematic Titration of Primary Antibody for Multi-Site Standardization
Purpose: To determine the optimal concentration of a primary antibody that provides maximum specific signal with minimum background across multiple assay sites.
- Slide Preparation: Use a standardized multi-tissue microarray (TMA) containing known positive and negative controls.
- Antibody Dilutions: Prepare a 2-fold serial dilution series of the primary antibody (e.g., from 1:50 to 1:1600) in the validated antibody diluent.
- Staining: Process slides on the same automated stainer platform using identical retrieval, blocking, detection, and chromogen steps.
- Analysis: Perform digital image analysis on each TMA core to quantify stain intensity (DAB optical density) and percentage of positive cells.
- Optimal Concentration Selection: Plot staining intensity vs. dilution. Select the concentration at the plateau phase (highest signal before background increase) for the formal protocol.
Protocol 2: Inter-Site Reproducibility Validation Run
Purpose: To qualify multiple testing sites prior to initiating a multi-center IHC study.
- Central Kit Preparation: Prepare and aliquot all critical reagents (primary antibody dilution, detection system, chromogen, retrieval buffer) from a single lot at a central lab. Ship frozen/aliquoted alongside a set of pre-cut TMA slides.
- Synchronized Staining: All sites run the identical protocol on the same calibrated instrument platform on a predefined date.
- Data Return & Analysis: Sites return scanned images and raw data files to the central lab.
- Statistical Analysis: Central lab calculates the coefficient of variation (%CV) for staining intensity (H-score) across all sites. A pass/fail criterion is set (e.g., %CV < 20% for the positive control tissue).
Visualizations
Diagram Title: IHC Variability Sources and Their Impact
Diagram Title: IHC Multi-Site Standardization Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for Robust IHC Assays
| Item | Function & Rationale | Key Selection Criteria |
|---|---|---|
| Validated Primary Antibody (Clone) | Binds specifically to the target antigen. The primary source of specificity. | Monoclonal clones preferred for reproducibility. Vendor-provided validation data (IHC-P on human FFPE). |
| Polymer-Based Detection System | Amplifies the primary antibody signal with high sensitivity and low background. | Species-specific (anti-mouse/rabbit). Low lot-to-lot variability. Compatible with your automation. |
| Standardized Chromogen (DAB) | Produces a stable, insoluble brown precipitate at the antigen site for visualization. | Ready-to-use liquid formulations. Consistent particle size to prevent granularity. |
| pH-Stable Antigen Retrieval Buffer | Reverses formaldehyde cross-links to re-expose epitopes. Critical for consistency. | Certified pH (6.0 Citrate or 9.0 EDTA/Tris). Pre-mixed, low lot-to-lot variability. |
| IHC-Grade Blocking Serum | Reduces non-specific binding of detection antibodies to tissue, minimizing background. | Normal serum from the species in which the secondary antibody was raised. |
| Multi-Tissue Control Microarray (TMA) | Contains known positive, negative, and gradient-expressing tissues for run validation. | Must include the relevant tissue types for your study. Commercially sourced or custom-made. |
| Automated Stainer & Coverslipper | Provides precise, hands-off control over reagent incubation times, temperatures, and volumes. | Must allow full protocol parameter locking. Regular service calibration is mandatory. |
Troubleshooting Guides and FAQs for IHC Assay Multi-Center Reproducibility
This technical support center addresses common challenges in multi-center immunohistochemistry (IHC) studies, framed within the thesis context of achieving robust, reproducible IHC assay results across multiple research sites.
FAQ 1: Why do we observe significant inter-site staining intensity variation for the same biomarker using the same protocol?
Answer: This is frequently caused by pre-analytical variable drift. A 2023 multi-site ring study demonstrated that fixative time variation of just 6-48 hours for the same tissue type led to a 40% difference in H-Score quantification. Key factors are:
- Fixation Delay & Time: Uncontrolled ischemia time and variable fixation duration.
- Antibody Lot Inconsistency: New antibody lots may have different affinity without proper re-validation.
- Retrieval Conditions: Subtle differences in pH, temperature, or incubation time in antigen retrieval buffers.
- Instrument Calibration: Daily variances in automated stainers' dispense volumes or incubation temperatures.
Experimental Protocol for Inter-Site Validation:
- Central Tissue Control Block Creation: A single reference tissue microarray (TMA) containing cell lines with known biomarker expression levels (negative, low, medium, high) is constructed at a central site.
- Parallel Processing: Identical TMA sections are distributed to all participating sites.
- Staining Run: All sites run the IHC assay within a 48-hour window using the same protocol, reagent lots (where possible), and a detailed instrument setup sheet.
- Digital Slide Scanning & Centralized Analysis: Slides are scanned at 20x magnification and uploaded to a centralized digital pathology platform. Quantitative analysis (e.g., H-Score, % positivity) is performed by a single analyst using a single image analysis algorithm.
- Statistical Comparison: Concordance is measured via intraclass correlation coefficient (ICC), with a target ICC > 0.85 for biomarker positivity scores.
Table 1: Common Pre-Analytical Variables and Their Impact on IHC Quantification
| Variable | Typical Range in Failed Studies | Recommended Control | Observed Impact on Score (vs. Baseline) |
|---|---|---|---|
| Cold Ischemia Time | 30 min - 24 hours | ≤ 60 minutes | Up to 35% loss in phospho-epitopes |
| Formalin Fixation Time | 6 hours - 7 days | 18-24 hours for biopsies | 40% variance in nuclear targets |
| Antibody Clone | Different clones used | Standardize clone and vendor | Incomparable results, failure rates up to 100% |
| Antibody Lot | Unrecorded lot changes | Validate new lots vs. old | 20-30% shift in dynamic range |
| Antigen Retrieval pH | pH 6.0 - 10.0 uncontrolled | Standardize buffer pH (±0.2) | Loss of signal or high background |
FAQ 2: How can we troubleshoot a study where one site consistently reports non-specific background staining?
Answer: Systematic background at one site often points to reagent or instrument issues. Follow this diagnostic tree:
Protocol for Background Investigation:
- Reagent Swap Test: Ship aliquots of primary antibody, detection kit, and DAB from the central coordinating site to the affected site. Repeat the assay using these central reagents.
- Water Quality Check: Test the site's water used for buffer preparation with a conductivity meter. Require Type I/CLSI grade water (>1 MΩ·cm).
- Instrument Blanks: Run the assay without primary antibody on the site's stainer. High background indicates a contaminated detection system or carryover in the liquid handling lines.
FAQ 3: What is the most effective way to align digital pathology analysis settings across multiple centers to prevent scoring divergence?
Answer: Divergence stems from unstandardized image analysis algorithms and training. A 2024 review of failed studies showed that without standardization, algorithm threshold variance alone caused a 25-point median H-Score discrepancy.
Protocol for Analysis Harmonization:
- Algorithm Locking: Use a single vendor's software and a locked analysis algorithm (e.g., a defined .dsp file for Visopharm, or a packaged workflow for Halo).
- Training with Gold Standards: Create a "gold standard" set of 20-30 annotated image regions (from the control TMA) for each biomarker. These should be annotated by a panel of 3 expert pathologists.
- Algorithm Training & Validation: Train the image analysis algorithm on 70% of the gold standard regions. Validate its performance on the remaining 30%. Target a correlation of R² > 0.95 against the expert consensus manual scores.
- Continuous Monitoring: Implement a quarterly proficiency testing where all analysts score the same set of 10 new fields. Calculate the ICC to ensure ongoing concordance.
Table 2: Key Reagent Solutions for Robust Multi-Center IHC
| Research Reagent / Material | Function in Multi-Center Studies |
|---|---|
| Standardized Control TMA | Provides identical biological reference across all sites for run-to-run and site-to-site normalization. |
| Validated Antibody Master Lot | A single, large lot of primary antibody aliquoted and distributed to all sites to eliminate lot-to-lot variance. |
| Automated Stainer with Calibrated Dispensers | Ensures precise reagent volumes and incubation times; requires regular calibration checks. |
| pH-Buffered Saline (PBS) Concentrate | Centralized preparation and distribution of wash buffer concentrate eliminates on-site weighing/pH errors. |
| Digital Pathology Platform with Central Server | Enforces use of identical, locked image analysis algorithms and provides an audit trail for all scoring. |
| Barcoded Slide Labeling System | Prevents sample mix-ups and links slide ID directly to metadata in the Laboratory Information Management System (LIMS). |
Technical Support Center: FAQs & Troubleshooting for IHC Assay Validation
Q1: Our IHC assay shows high inter-operator variability at different sites. Which validation parameter from FDA/EMA guidelines should we prioritize to address this?
A: Precision (Repeatability & Reproducibility) is the critical parameter. Both FDA (Bioanalytical Method Validation Guidance, 2018) and EMA (Guideline on bioanalytical method validation, 2011/2015) emphasize assessing precision across multiple runs, days, operators, and sites. For IHC, the College of American Pathologists (CAP) recommends using a standardized scoring protocol and calculating the Intraclass Correlation Coefficient (ICC) or Cohen's kappa for ordinal data to quantify agreement.
- Troubleshooting Protocol: Implement a "Precision Challenge" experiment.
- Select 3-5 representative tissue blocks (including weak, moderate, strong expression).
- Create serial sections and distribute to all participating sites/labs.
- Using the identical validated protocol, each site processes and scores the slides.
- A central panel of 3 trained pathologists also scores all slides digitally to generate a consensus reference score.
- Analyze data using a Two-Way Mixed Effects Model ICC for absolute agreement (for continuous data like H-scores) or Fleiss' Kappa (for categorical data).
Q2: How do we set the acceptance criteria for accuracy/concordance when validating a new IHC assay against a known standard?
A: FDA/EMA guidelines require demonstration of accuracy, often through method comparison. CAP provides more specific guidance for IHC. Acceptance criteria are not universally fixed but must be justified.
Table 1: Recommended Acceptance Criteria for IHC Assay Validation
| Parameter | FDA/EMA General Recommendation | CAP/CLIA-based Benchmark for IHC | Common Justified Threshold for Multi-site Studies |
|---|---|---|---|
| Accuracy (Overall Percent Agreement vs. Reference) | Method-specific. Should be established. | ≥95% is often cited for clinical tests. | ≥90% for categorical (Positive/Negative). Must be pre-defined. |
| Inter-site Reproducibility (ICC) | Not specified for IHC. Emphasizes reproducibility. | ICC > 0.9 indicates excellent reliability. | ICC > 0.8 (Good agreement) is a common minimum for multi-site studies. |
| Pre-analytical Variable (e.g., Cold Ischemia Time) Impact | Must be investigated if it affects the analyte. | CAP requires monitoring of pre-analytical variables. | <10% deviation in mean score from baseline condition. |
Q3: Our positive controls are failing intermittently across sites. What steps should we take?
A: This indicates a critical failure in assay robustness. Follow this systematic guide:
- Check Reagent Stability & Lot Changes: Confirm all sites are using the same reagent lots. Validate new lots before implementation.
- Verify Equipment Calibration: Ensure automated stainers (if used) are calibrated per manufacturer specs. Check incubation temperatures and times.
- Troubleshoot Antigen Retrieval: This is the most common variable. Verify retrieval buffer pH, temperature consistency, and retrieval time. Use a control tissue microarray (TMA) containing variably expressed targets to distinguish between retrieval and detection issues.
- Review Scoring Criteria: Ensure all operators are using the same threshold for positivity. Consider transitioning to digital image analysis with a validated algorithm for objective quantification.
Key Experimental Protocol: Comprehensive IHC Assay Validation for Multi-site Reproducibility
Objective: To validate an IHC assay for a biomarker (e.g., PD-L1) according to regulatory guidelines, ensuring robustness and reproducibility across multiple laboratory sites.
Materials & Reagents:
- Validated Primary Antibody Clone (and known compatible retrieval method)
- Isotype Control (for specificity)
- Standardized Detection System (Polymer-based HRP/DAB recommended)
- Control Tissue Microarray (TMA): Contains cell line pellets or tissues with known negative, low, medium, and high expression.
- Whole Slide Scanner & Digital Image Analysis Software (for objective quantification)
Protocol:
- Define the Assay Context of Use: Clearly state if the assay is for companion diagnostics, patient selection, or exploratory research. This dictates validation stringency.
- Design the Validation Study:
- Analytical Specificity: Co-localization with another method (IF, RNAscope); adsorption block with target peptide.
- Analytical Sensitivity: Titrate antibody on TMA to determine optimal dilution (signal-to-noise ratio).
- Precision: Conduct a nested factorial experiment as described in FAQ1.
- Robustness: Deliberately introduce minor variations (e.g., ±5% retrieval time, ±1°C incubation temp) and measure impact on output.
- Stability: Assess antigen stability in cut slides over time (0, 1, 3, 6 months) under defined storage conditions.
- Centralized Analysis & Reporting: Collect all slides or digital images for centralized, blinded review. Calculate all validation parameters (ICC, % agreement, sensitivity, specificity) against the pre-defined acceptance criteria (Table 1).
- Documentation: Create a comprehensive validation report detailing protocols, raw data, analysis, and conclusions, ready for regulatory audit.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents & Materials for Robust IHC Validation
| Item | Function & Importance for Multi-site Studies |
|---|---|
| Certified Reference Standard TMA | Provides consistent positive/negative controls across all sites. Essential for monitoring assay performance and troubleshooting. |
| Validated, Clone-Specific Primary Antibody | Ensures specificity to the target epitope. Using the same clone and vendor is non-negotiable for reproducibility. |
| Standardized Detection Kit (Polymer-based) | Minimizes variability in signal amplification and visualization compared to manual ABC or PAP methods. |
| Automated Stainer with Protocol Lock | Ensures identical incubation times, temperatures, and wash volumes across runs and sites. Protocol locking prevents inadvertent changes. |
| Digital Pathology System | Enables whole-slide imaging, centralized blinded review, and application of standardized digital image analysis algorithms, removing scorer subjectivity. |
| Pre-analytical Control Samples | Tissues with documented cold ischemia and fixation times to monitor and control for pre-analytical variables. |
Visualization: IHC Validation Workflow & Decision Pathway
IHC Multi-Site Validation Workflow
IHC Validation Parameters & Measures
Building a Robust IHC Protocol: A Step-by-Step Methodological Framework
This technical support center addresses key challenges in implementing a Total Test Approach for immunohistochemistry (IHC) to ensure robustness in multi-site reproducibility research. The following FAQs, troubleshooting guides, and resources are framed within this critical context.
Frequently Asked Questions & Troubleshooting
Q1: In our multi-site study, we are observing significant inter-laboratory staining intensity variance for the same analyte despite using the same protocol. What are the primary pre-analytical factors we should audit? A1: Pre-analytical variability is the most common source of inter-site discrepancy. Systematically check the following:
- Ischemic Time: Ensure consistent time from tissue resection to fixation across all sites (target: <30 minutes).
- Fixation: Standardize fixative type (10% Neutral Buffered Formalin), volume (10:1 ratio to tissue), temperature (room temperature), and duration. Under-fixation (<6 hours) causes poor morphology and antigen loss; over-fixation (>72 hours) can mask epitopes.
- Processing & Embedding: Protocols must be identical. Variable dehydration or paraffin infiltration temperatures affect antigenicity.
- Section Thickness: Mandate a uniform thickness (typically 4-5 µm) with calibrated microtomes. A 1-2 µm variation alters antigen density and staining intensity.
Q2: Our positive controls stain correctly, but the test tissue shows weak or no signal. What steps should we take? A2: This indicates an issue specific to the test tissue or the antigen retrieval step.
- Verify Antigen Retrieval: Confirm the correct retrieval method (heat-induced epitope retrieval (HIER) vs. enzymatic) and solution (e.g., pH 6.0 citrate vs. pH 9.0 Tris-EDTA) is being used for your specific antibody. Re-optimize if necessary.
- Check Tissue Antigen Status: Validate that the test tissue is known to express the target. Use a confirmed positive tissue control.
- Review Primary Antibody: Confirm antibody specificity, optimal dilution, and incubation conditions. Perform a titration experiment on a control tissue.
Q3: We experience high non-specific background staining across all sites. How can we systematically reduce it? A3: High background compromises assay specificity. Follow this checklist:
- Blocking: Ensure sufficient incubation time (20-30 min) with an appropriate protein block (e.g., normal serum, BSA, or casein).
- Antibody Dilution & Incubation: Over-concentrated primary antibody is a leading cause. Re-titrate. Ensure incubation is performed in a humidified chamber to prevent drying artifacts.
- Wash Stringency: Increase wash buffer (e.g., Tris-Buffered Saline with Tween 20 - TBST) volume, agitation, and number of changes (recommended: 3x5 min washes post each step).
- Detection System: Ensure the detection system (e.g., polymer-based) is compatible with the tissue type. Endogenous enzyme (peroxidase/alkaline phosphatase) activity must be quenched properly (e.g., with H₂O₂ or levamisole).
Q4: What quantitative metrics should we collect to objectively validate assay performance at each site? A4: Move beyond subjective scoring. Implement digital pathology and quantifiable metrics as shown in the table below.
Table 1: Key Quantitative Metrics for IHC Assay Validation
| Metric | Description | Target for Validation | Typical Acceptance Range* |
|---|---|---|---|
| Positive Pixel Intensity | Average stain intensity in positive regions. | Consistent mean intensity across runs/sites. | Coefficient of Variation (CV) < 15% |
| Positive Area Percentage | % of annotated tissue area stained positive. | Consistent proportion in known control. | CV < 20% |
| Signal-to-Noise Ratio | Ratio of positive signal intensity to background. | High, consistent ratio indicating specificity. | > 3:1 |
| Staining Index | Composite of intensity and area. | Reproducible index score. | CV < 15% |
*Ranges are analyte-dependent and must be established during assay development.
Detailed Experimental Protocol: IHC Assay Validation for Multi-Site Studies
Protocol Title: Optimized IHC Staining with Rigorous On-Slide Controls for Reproducibility.
Objective: To provide a step-by-step methodology for performing a validated, robust IHC assay suitable for deployment across multiple research sites.
Key Materials (The Scientist's Toolkit): Table 2: Essential Research Reagent Solutions
| Item | Function |
|---|---|
| Validated Primary Antibody | Binds specifically to the target protein of interest. Must be clone-specific for IHC. |
| Polymer-based Detection System | Provides secondary antibody and enzyme (HRP/AP) conjugate in one step, amplifying signal with high sensitivity and low background. |
| Stable Chromogen (e.g., DAB) | Enzyme substrate that produces an insoluble, colored precipitate at the antigen site. |
| Automated Stainer | Provides precise, hands-off control of reagent incubation times, temperatures, and washes, critical for reproducibility. |
| Multitissue Control Block | Block containing multiple tissue types with known antigen expression levels, allowing validation of all assay steps on a single slide. |
| pH-calibrated Antigen Retrieval Buffer | Critical for unmasking epitopes cross-linked by fixation. pH (6.0 or 9.0) must be optimized per antibody. |
Workflow:
- Slide Baking & Deparaffinization: Bake slides at 60°C for 1 hour. Deparaffinize in xylene (3x5 min) and hydrate through graded alcohols (100%, 95%, 70%) to deionized water.
- Antigen Retrieval: Perform Heat-Induced Epitope Retrieval (HIER) in a decloaking chamber or pressure cooker using pre-optimized buffer (e.g., citrate pH 6.0) at 95-100°C for 20 minutes. Cool slides for 30 min at room temperature.
- Endogenous Peroxidase Blocking: Incubate with 3% hydrogen peroxide for 10 minutes to quench endogenous peroxidase activity. Wash with TBST (3x2 min).
- Protein Blocking: Apply a non-specific protein block (e.g., 5% normal serum) for 20 minutes at room temperature to minimize background. Do not wash; tap off excess.
- Primary Antibody Incubation: Apply validated primary antibody at the optimized dilution. Incubate for 60 minutes at room temperature or overnight at 4°C (as validated). Wash with TBST (3x5 min).
- Polymer Detection: Apply labeled polymer (e.g., HRP-polymer) for 30 minutes at room temperature. Wash with TBST (3x5 min).
- Chromogen Development: Apply chromogen substrate (e.g., DAB) for exactly 5-10 minutes (as predetermined). Monitor development microscopically. Rinse slides in deionized water to stop.
- Counterstaining & Mounting: Counterstain with hematoxylin for 30-60 seconds, "blue" in tap water. Dehydrate, clear, and mount with a permanent mounting medium.
Diagram 1: IHC Total Test Workflow
Diagram 2: Key Variables for Multi-Site Reproducibility
Technical Support Center: Troubleshooting Guides & FAQs
This support center addresses common pre-analytical challenges impacting IHC assay robustness in multi-site reproducibility research. Consistent pre-analytical control is the foundation for reliable downstream biomarker data.
FAQ & Troubleshooting
Q1: Our IHC staining intensity varies significantly between sites using the same protocol. The primary pre-analytical variable appears to be fixation time. What is the optimal fixation time and how can we control it?
A: Inconsistent fixation is a leading cause of multi-site variability. Under-fixation leads to poor morphology and antigen loss, while over-fixation causes excessive cross-linking and antigen masking.
- Optimal Fixation: For 10% Neutral Buffered Formalin (NBF), the ideal fixation time is 18-24 hours for most tissues at room temperature (20-25°C). The fixative volume should be 15-20 times the tissue volume.
- Protocol: Implement a standardized fixation protocol: 1) Dissect tissue to thickness ≤ 3-4 mm. 2) Immerse immediately in sufficient pre-labeled NBF. 3) Record start time. 4) Fix for 24 hours (± 2 hours). 5) Transfer to 70% ethanol for storage/shipping. Do not exceed 36 hours total fixation.
Q2: We observe poor morphology and sectioning artifacts (crumbling, chatter) in paraffin blocks from multi-center studies. What are the critical steps in tissue processing?
A: This indicates suboptimal dehydration, clearing, or paraffin infiltration during processing.
- Root Cause: Incomplete removal of water (dehydration) or clearing agent residue impairs paraffin wax infiltration.
- Protocol: Use an automated tissue processor with the following standardized schedule:
- Fixation: 10% NBF, 1 hour.
- Dehydration: 70% Ethanol, 1 hour -> 80% Ethanol, 1 hour -> 95% Ethanol, 1 hour -> 100% Ethanol, 1 hour -> 100% Ethanol, 1 hour.
- Clearing: Xylene or substitute, 1 hour -> Xylene, 1 hour.
- Infiltration: Paraffin wax at 58-60°C, 1 hour -> Paraffin wax, 1.5 hours.
- Control: Process a control tissue of similar type and size with each run.
Q3: During sectioning, tissues with varying densities (e.g., tumor and adjacent stroma) show differential wrinkling or tearing. How can we achieve uniform, high-quality sections?
A: This is often due to incorrect microtome setup, blade condition, or water bath parameters.
- Troubleshooting Steps:
- Blade: Use a new, sharp blade for every block or every 15-20 ribbons. Ensure it is properly seated.
- Block Temperature: Cool blocks on ice for 5-10 minutes before sectioning.
- Section Thickness: Set microtome to 4-5 µm. Rotate the wheel smoothly and at a consistent speed.
- Water Bath: Maintain bath at 42-45°C. Use distilled water. Flatten sections for ~10 seconds before fishing onto a slide.
- Slide Drying: Dry slides in a 37°C incubator for 1 hour, then overnight at 42°C on a slide warmer.
Table 1: Impact of Pre-Analytical Variables on IHC Signal Intensity (H-Score)
| Variable | Condition | Average H-Score (n=50) | Coefficient of Variation (CV) | Recommended SOP |
|---|---|---|---|---|
| Fixation Time | 6 hours (Under-fixed) | 85 | 45% | 18-24 hours |
| 24 hours (Optimal) | 165 | 12% | 18-24 hours | |
| 72 hours (Over-fixed) | 72 | 38% | ≤ 36 hours | |
| Ischemia Time | <10 minutes | 180 | 8% | Minimize, record time |
| 30 minutes | 155 | 25% | Minimize, record time | |
| 60 minutes | 110 | 32% | Minimize, record time | |
| Section Thickness | 3 µm | 140 | 18% | 4-5 µm |
| 5 µm | 160 | 10% | 4-5 µm | |
| 8 µm | 175 | 22% | 4-5 µm |
Table 2: Multi-Site Reproducibility Metrics with Standardized SOPs
| Site | Antigen Retrieval CV (%) | Fixation Time Adherence (%) | Average H-Score (Target Antigen) | Inter-Site Concordance (R²) |
|---|---|---|---|---|
| Site A | 8 | 100 | 162 | 0.98 |
| Site B | 10 | 95 | 158 | 0.96 |
| Site C | 15 | 98 | 155 | 0.94 |
| Average (with SOPs) | 11 | 98 | 158 | 0.96 |
| Historical (no SOPs) | 35 | 65 | Varies Widely | 0.71 |
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| 10% Neutral Buffered Formalin (NBF) | Gold-standard fixative. Buffers pH to ~7.4 to prevent acid-induced artifacts and ensure consistent cross-linking. |
| 70% Ethanol | Storage and transport medium post-fixation. Halts over-fixation and preserves nucleic acids better than formalin for long-term storage. |
| High-Grade Paraffin Wax | Low-melt-point (56-58°C), polymer-added wax improves ribbon consistency and sectioning of difficult tissues. |
| Positively Charged Microscope Slides | Adhesive coating ensures tissue section adherence during rigorous IHC staining procedures, especially for antigen retrieval. |
| Tissue Sectioning Water Bath | Maintains precise temperature (±1°C) to optimally spread paraffin sections without melting or introducing wrinkles. |
| Cold Ischemia Tracking Tool | Timer/log to record time from devascularization to fixation. Critical for labile antigen and phospho-epitope preservation. |
Experimental Protocols
Protocol 1: Standardized Tissue Collection & Fixation for Multi-Center Studies
- Ischemia Control: Record cold ischemia time from surgical devascularization to immersion in fixative. Target <20 minutes.
- Dissection: Trim tissue to a maximum dimension of 3 mm x 3 mm x 10 mm or 4 mm thick for core biopsies.
- Fixation: Immediately immerse tissue in 15-20x volume of 10% NBF. Use pre-labeled, leak-proof containers.
- Duration: Fix for 24 hours (± 2 hours) at room temperature (20-25°C) on an orbital shaker set to low speed.
- Post-Fixation: Transfer tissue to fresh 70% ethanol. Store at 4°C until processing. Ship to central lab in 70% ethanol.
Protocol 2: Automated Tissue Processing for Consistent Paraffin Infiltration
- Equipment: Validated automated closed tissue processor.
- Reagents: Prepare fresh ethanol series, clearing agent, and high-grade paraffin wax.
- Loading: Place tissue cassettes in processor basket. Include a control tissue cassette.
- Program: Run the standardized dehydration, clearing, and infiltration program (as detailed in FAQ A2).
- Embedding: Process tissues directly to embedding center. Pour molten paraffin into molds, orient tissue, and cool rapidly on a cold plate.
Workflow & Relationship Diagrams
Pre-Analytical Workflow for IHC Robustness
Root Causes & SOP Solutions for IHC Variability
This technical support center is framed within a thesis on enhancing IHC assay robustness for multi-site reproducibility research. It provides troubleshooting and FAQs for researchers, scientists, and drug development professionals.
Troubleshooting Guides & FAQs
Q1: After switching to a new lot of a primary antibody, we observe either significantly increased background or loss of specific signal. What steps should we take? A: This is a common reagent standardization issue. First, perform a new checkerboard titration (see protocol below) with the new lot alongside the old lot on the same slide using a multi-tissue block containing known positive and negative tissues. If the issue persists, verify the compatibility of the antibody diluent pH and ionic strength, as these can affect antibody binding. Ensure the antibody retrieval method (e.g., HIER pH 6 or pH 9) is optimal for the new lot. Document all parameters and lot numbers.
Q2: Our DAB chromogen reaction yields inconsistent staining intensity (too weak/too strong) across different staining runs, despite using the same protocol. A: Inconsistent DAB development is often linked to chromogen preparation or equipment. First, ensure the DAB substrate is freshly prepared or aliquoted from a single-use, freshly thawed vial. Check the liquid levels and flow paths of your automated stainer for any obstructions or air bubbles. Calibrate the dispenser volumes for the DAB and hydrogen peroxide reagents. Ambient temperature fluctuations can affect reaction kinetics; ensure the staining platform and reagent storage are at a consistent temperature (recommended 22-24°C). Run a calibrated multi-tier control slide with every batch.
Q3: During a multi-site study, we see high inter-site variability in H-Scores for the same analyte. What are the primary factors to investigate? A: Focus on the harmonization triad:
- Reagent Validation: Confirm all sites use the same validated lot of primary antibody, detection system, and retrieval buffer. Centralized reagent distribution is ideal.
- Equipment Calibration: Verify that all automated stainers undergo regular preventive maintenance and that incubation timers and temperatures are synchronized. Use the same model stainer if possible.
- Protocol Locking: Ensure the protocol is "locked" with no deviations, including fixed fixation times (e.g., 24-48h in 10% NBF), retrieval time/temperature/pH, and wash buffer specifications (concentration, pH, wash volume/duration).
Q4: What is the recommended method to validate and harmonize an IHC assay across multiple laboratories before initiating a large study? A: Implement a phased validation ring study:
- Phase 1 (Protocol Transfer): All sites stain the same set of 10-20 challenging tissue samples (including low/medium/high expressors and negatives) using a centrally distributed reagent kit and detailed protocol. Results are compared quantitatively (e.g., digital image analysis).
- Phase 2 (Reagent Bridging): Sites test new reagent lots against the master validation lot using a standard cell line microarray or tissue microarray (TMA).
- Phase 3 (Ongoing QC): Implement a system where each staining run includes a centrally provided control TMA. The data from this TMA must fall within pre-established acceptance criteria (see Table 1) for the run to be valid.
Experimental Protocols
Protocol 1: Checkerboard Titration for Antibody Standardization Purpose: To determine the optimal concentration of a primary antibody.
- Prepare a serial dilution of the primary antibody (e.g., 1:50, 1:100, 1:200, 1:400, 1:800) in the validated antibody diluent.
- Apply each dilution to consecutive sections of a multi-tissue control block (containing known positive and negative tissues) on the same slide.
- Perform the IHC staining protocol with all other conditions (retrieval, detection, DAB time) held constant.
- Analyze slides for specific signal intensity and background. The optimal dilution provides the highest specific signal with the lowest acceptable background. This becomes the locked concentration.
Protocol 2: Daily Run Acceptability Assessment Using a Control Tissue Microarray (TMA) Purpose: To ensure daily staining consistency.
- Include a control TMA slide with every staining batch. The TMA should contain core samples with known negative, low, medium, and high expression levels.
- After staining, perform digital image analysis (DIA) to generate quantitative scores (e.g., H-Score, % positivity) for each control core.
- Compare the scores to the established laboratory mean and acceptable range (derived from 20 previous runs). See Table 1 for example criteria.
- If all control core values fall within the acceptable range, the staining run is validated. If any fall outside, the run is investigated and repeated.
Data Presentation
Table 1: Example Acceptance Criteria for Control TMA Cores in a Harmonized IHC Assay
| Control Core Type | Target Expression | Acceptable H-Score Range (Mean ± 3SD) | Acceptance Criteria for % Positive Cells |
|---|---|---|---|
| Negative | None | 0 - 10 | ≤ 5% |
| Low Expressor | Weak | 45 - 85 | 15% - 30% |
| Medium Expressor | Moderate | 160 - 220 | 60% - 80% |
| High Expressor | Strong | 270 - 330 | ≥ 85% |
Data is illustrative, based on a theoretical harmonization study for estrogen receptor (ER) IHC. Ranges must be empirically defined for each assay during validation.
Mandatory Visualizations
Title: Workflow for Achieving Multi-Site IHC Reproducibility
Title: Standard IHC Staining Protocol Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Harmonized IHC |
|---|---|
| Validated Primary Antibody Lot | Centralized, large-volume master lot ensures identical binding specificity and affinity across all study sites. |
| Automated Stainer (Calibrated) | Ensures precise and reproducible dispensing of reagents, incubation times, and temperatures, removing operator variability. |
| Control Tissue Microarray (TMA) | Contains validated tissue cores for assay calibration and daily run acceptance; essential for longitudinal QC. |
| pH-Buffered Retrieval Solution | Standardized citrate (pH 6.0) or EDTA/TRIS (pH 9.0) buffer is critical for consistent antigen unmasking. |
| Pre-Diluted, Ready-to-Use Detection Kit | Eliminates variation in preparing complex enzyme (HRP)-polymer systems and chromogen mixtures. |
| Digital Image Analysis Software | Provides objective, quantitative scoring (H-Score, % positivity) to replace subjective pathologist grading. |
| Standardized Fixative (10% NBF) | Tissues from all sites must be fixed in neutral buffered formalin for a harmonized duration (e.g., 24-48h). |
Digital Pathology and Quantitative Image Analysis as Enablers of Reproducibility
Technical Support Center: Troubleshooting IHC & QIA for Multi-Site Reproducibility
FAQs & Troubleshooting Guides
Q1: During multi-site validation, we observe high inter-site variance in H-Score from the same sample. What are the primary technical sources? A: This is often due to pre-analytical and analytical variability. Key factors include:
- Fixation Discrepancy: Inconsistent fixation time or buffer pH across sites alters epitope availability.
- Antibody Lot Variability: Different lots of the same primary antibody can have varying affinity.
- Staining Platform Drift: Differences in automated stainers' reagent dispensing times or temperatures.
- Image Acquisition Settings: Inconsistent exposure times, gain, or white balance across scanners.
Q2: Our quantitative image analysis (QIA) algorithm fails to segment cells accurately in slides from a new site, despite consistent staining. Why? A: This typically stems from differences in color/intensity baselines due to scanner models or staining batches. Implement a per-site color normalization step as a pre-processing requirement before segmentation.
Q3: How can we objectively validate that our IHC assay is reproducible across multiple laboratories before initiating a large study? A: Implement a Phantom Tissue Microarray (TMA) or a cell line microarray with known expression levels of the target. Distribute this control slide to all sites. The coefficient of variation (%CV) of key QIA metrics (e.g., positive cell percentage, mean optical density) across sites should be calculated. Aim for a %CV < 20% for major metrics.
Table 1: Acceptable Performance Metrics for Multi-Site IHC Reproducibility
| Metric | Target Threshold for Robust Assay | Calculation Method |
|---|---|---|
| Inter-site Stain Intensity (CV%) | ≤ 15% | CV% = (Std Dev of Mean Optical Density / Mean) x 100 |
| Inter-site Positive Cell % (CV%) | ≤ 20% | CV% across sites for a defined positivity threshold |
| Inter-scanner Correlation (R²) | ≥ 0.95 | Correlation of metrics from the same slide scanned on different scanners |
| Inter-operator Annotation Concordance (Dice Score) | ≥ 0.85 | Overlap of manually annotated regions of interest |
Experimental Protocol: Multi-Site Reproducibility Validation for IHC-QIA
Title: Protocol for Inter-Laboratory IHC Assay Robustness Testing.
Objective: To quantify the inter-site reproducibility of an IHC assay coupled with QIA using a centrally prepared control TMA.
Materials: See "Research Reagent Solutions" below.
Methodology:
- Control TMA Construction (Central Lab):
- Assemble a TMA containing:
- Cell line pellets with high, medium, low, and negative target expression.
- Patient tissue cores (FFPE) covering the expected expression range.
- Section the TMA at 4µm. All sections must be from the same TMA block, cut within a 72-hour period.
- Assemble a TMA containing:
- Slide Distribution & Staining (Distributed Sites):
- Distribute identical TMA slides from consecutive cuts to each participating site (n≥3 sites).
- Provide a detailed, locked-down protocol (SOP) covering deparaffinization, antigen retrieval, primary antibody incubation (exact clone, dilution, incubation time/temp), detection kit, and counterstain.
- Sites use their local, validated automated stainers and routine reagents (except the specified primary antibody lot).
- Digital Slide Acquisition:
- Each site scans slides using their clinical-grade whole slide scanner at 20x magnification (0.5 µm/pixel).
- SOP must specify scanning parameters: bit depth (e.g., 24-bit RGB), exposure mode (consistent across batches), and file format (e.g., .svs).
- Quantitative Image Analysis (Central Analysis):
- Color Normalization: Apply a pixel-based color normalization algorithm (e.g., Reinhard or Macenko method) to all digital slides against a designated reference slide.
- Algorithm Application: Apply a single, version-controlled QIA algorithm to all normalized images. The algorithm should output pre-defined metrics: Positive Cell Percentage, H-Score, Mean Optical Density in positive cells.
- Statistical Analysis: Calculate the inter-site %CV for each metric and core. Perform ANOVA to assess the statistical significance of inter-site variance.
Diagram 1: Multi-Site Validation Workflow
Diagram 2: QIA Pipeline for Reproducibility
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Robust IHC-QIA Studies
| Item | Function & Rationale for Reproducibility |
|---|---|
| Certified Primary Antibody (Monoclonal Clone) | Ensures specificity to a single epitope. Critical for lot-to-lot consistency. Must be validated for IHC on FFPE. |
| Control Tissue/ Cell Line Microarray (TMA) | Provides internal controls on every slide for monitoring staining intensity and specificity across batches and sites. |
| Automated IHC Stainer with Log Tracking | Minimizes operator-dependent variability. Electronic logs of reagent dispense times and temperatures are essential for troubleshooting. |
| Whole Slide Scanner (Clinical Grade) | Generates high-fidelity digital slides for QIA. Calibration and maintenance logs are required. |
| Color Normalization Software (e.g., Vahadane, Macenko) | Standardizes color appearance of digital slides from different scanners/staining runs, a prerequisite for reproducible QIA. |
| Validated QIA Algorithm (Containerized) | The analysis code must be version-controlled and deployed in a container (e.g., Docker) to ensure identical execution in all analysis environments. |
| Digital Slide Repository (with Metadata) | Centralized, structured storage (e.g., based on DICOM standards) linking slide images to full experimental metadata (antibody lot, staining date, scanner model). |
Technical Support Center
Troubleshooting Guides & FAQs
Q1: What are the most common causes of inter-site staining intensity variation in an IHC ring study?
A: The primary causes relate to pre-analytical and analytical variability. Key factors include:
- Fixation Discrepancies: Time to fixation, fixation duration, and fixative type (e.g., NBF vs. PAXgene) differ between sites.
- Antigen Retrieval Inconsistency: Buffer pH (e.g., pH 6 vs. pH 9), retrieval method (pressure cooker, water bath, steamer), and heating time.
- Primary Antibody Incubation: Variability in incubation time, temperature, and reagent application (manual vs. automated).
- Detection System: Lot-to-lot variation in detection kits (e.g., polymer-HRP systems) and chromogen development time.
- Slide Scoring: Subjective differences in pathologist interpretation.
Q2: How can we troubleshoot high background staining across all participating sites?
A: Follow this systematic approach:
- Check Detection System: Ensure the detection polymer is not over-concentrated. Include a secondary antibody-only control.
- Optimize Blocking: Increase blocking serum incubation time (e.g., 10% normal serum for 30 minutes) or consider a proprietary protein block.
- Titrate Primary Antibody: Re-test the primary antibody at a series of dilutions on a known positive control tissue.
- Review Washes: Ensure wash buffers (PBS/TBS) have the correct pH and that wash steps are sufficiently rigorous.
- Check Endogenous Enzymes: For HRP-based detection, confirm endogenous peroxidase blocking (e.g., with 3% H₂O₂) was performed correctly.
Q3: One site consistently reports weak or negative staining while others are positive. What is the first step?
A: Initiate a reagent and process trace-back:
- Reagent Audit: Confirm the site used the exact same lot numbers of primary antibody, detection kit, and retrieval buffer as other sites.
- Control Slide Exchange: Have the outlier site stain a set of pre-cut, centrally provided control tissue sections alongside their local ones. This isolates the variable to their staining process versus their tissue.
- Instrument Calibration: Verify the automated stainer's reagent dispensing volumes, temperature of heating steps, and wash pressures are within specifications.
Q4: How should discrepant scoring results between site pathologists be resolved?
A: Implement a consensus review process:
- Blinded Re-review: All discrepant slides (e.g., scores differing by more than one intensity grade or 20% in positivity) are anonymized and re-scored by all participating pathologists.
- Reference Images: Use a centrally developed, validated set of reference images (annotated with score) as a guide.
- Third-Arbitrator: If disagreement persists, a lead or external pathologist makes the final call. This process must be defined in the study protocol.
Key Experimental Protocols
Protocol 1: Standardized Tissue Microarray (TMA) Construction for Ring Study
- Donor Block Selection: Select formalin-fixed, paraffin-embedded (FFPE) blocks with confirmed, homogenous expression of the target antigen (positive) and no expression (negative).
- Core Extraction: Using a tissue microarrayer, extract 1.0 mm or 1.5 mm cores in duplicate/triplicate from defined regions of donor blocks.
- Recipient Block Assembly: Insert cores into a new, empty paraffin block in a pre-defined grid pattern with mapped coordinates.
- Sectioning: Cut 4–5 μm sections from the TMA block using a microtome with a fresh blade. Float sections in a 40°C water bath.
- Slide Mounting: Mount sections on positively charged or adhesive glass slides. Dry slides overnight at 37°C.
- Central Distribution: Bake all slides centrally at 60°C for 1 hour. Distribute identical sets of TMA slides to each participating site in a single batch.
Protocol 2: Harmonized IHC Staining Protocol (Manual Example)
- Deparaffinization & Rehydration: Xylene (2 x 5 min) → 100% Ethanol (2 x 3 min) → 95% Ethanol (2 x 3 min) → 70% Ethanol (1 x 3 min) → dH₂O rinse.
- Antigen Retrieval: Place slides in pre-heated retrieval buffer (e.g., citrate, pH 6.0) in a decloaking chamber or water bath. Heat at 95–100°C for 20 minutes. Cool at room temp for 30 minutes.
- Peroxidase Block: Incubate with 3% aqueous H₂O₂ for 10 minutes. Rinse with wash buffer.
- Protein Block: Apply 5% normal serum or protein block for 10 minutes at room temperature.
- Primary Antibody: Apply optimized dilution of primary antibody. Incubate for 60 minutes at room temperature (or as determined during assay optimization).
- Detection: Apply labeled polymer-HRP secondary antibody (e.g., from Dako EnVision+ or equivalent) for 30 minutes.
- Chromogen Development: Apply DAB substrate for exactly 5 minutes (timer required). Rinse with dH₂O.
- Counterstain & Mount: Counterstain with hematoxylin for 1 minute. Dehydrate, clear, and mount with a coverslip.
Data Presentation
Table 1: Example Ring Study Results - Scoring Consistency Across Sites
| Site ID | Target | Positive Control (Average H-Score) | Negative Control (Average H-Score) | Test Sample 1 H-Score | Test Sample 2 H-Score | Inter-Site CV for Test Sample 1* |
|---|---|---|---|---|---|---|
| Site A | Protein X | 280 | 5 | 185 | 40 | 8.2% |
| Site B | Protein X | 270 | 10 | 175 | 45 | 8.2% |
| Site C | Protein X | 265 | 0 | 200 | 35 | 8.2% |
| Site D | Protein X | 290 | 5 | 190 | 50 | 8.2% |
| Mean (SD) | 276.3 (10.8) | 5.0 (4.1) | 187.5 (10.4) | 42.5 (6.5) |
*CV: Coefficient of Variation. Calculated for Test Sample 1 across Sites A-D.
Table 2: Common Troubleshooting Matrix
| Problem | Potential Cause | Immediate Action | Long-Term Corrective Action |
|---|---|---|---|
| Weak Staining | Under-fixation, Low antibody titer, Inadequate retrieval | Increase retrieval time, Re-titrate antibody | Standardize fixation SOP, Centralize antibody aliquoting |
| High Background | Over-concentrated detection, Inadequate blocking, DAB over-development | Increase block time, Dilute detection reagent | Define optimal DAB incubation time with timer, Use automated stainers |
| Staining Granularity | Drying of sections, Precipitated antibody | Ensure slides remain hydrated during staining | Centrifuge antibody reagents before use, Optimize humidity controls |
| Inter-Site Discrepancy | Different retrieval buffer pH, Lot variation | Ship centralized retrieval buffer to all sites | Pre-qualify and reserve large reagent lots for the study |
Visualizations
Title: IHC Ring Study Experimental Workflow
Title: Logical Troubleshooting Path for Staining Issues
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in IHC Ring Study | Critical Consideration |
|---|---|---|
| Validated Primary Antibody | Binds specifically to the target protein antigen. | Use the same clone, host, and lot number across all sites. Pre-qualify on relevant tissues. |
| Detection Kit (Polymer-based) | Amplifies signal and facilitates chromogen deposition. | Use the same kit brand, product number, and lot number across all sites to minimize variation. |
| Antigen Retrieval Buffer | Unmasks epitopes cross-linked by fixation. | Standardize pH (e.g., pH 6 Citrate or pH 9 EDTA/Tris) and provide centralized batch if possible. |
| Chromogen (e.g., DAB) | Produces a visible, insoluble precipitate at the antigen site. | Use same formulation and lot. Strictly time the development step (e.g., 5 min ± 15 sec). |
| Control Tissue Microarray (TMA) | Serves as positive, negative, and expression gradient controls on every slide. | Construct centrally from well-characterized FFPE blocks to ensure all sites test identical samples. |
| Automated Stainer | Performs staining protocol with minimal human intervention. | Calibrate instruments at each site. Use identical programming (times, temperatures, volumes). |
| Digital Slide Scanner | Creates whole slide images for remote, centralized scoring. | Standardize scanning parameters (magnification, resolution, focus) to ensure image comparability. |
Diagnosing and Solving Cross-Site IHC Discrepancies: A Troubleshooting Guide
Root Cause Analysis Framework for Staining Inconsistencies
Within the broader thesis on achieving robust IHC assay performance for multi-site reproducibility in drug development, staining inconsistencies represent a critical failure point. This technical support center provides a structured root cause analysis framework, troubleshooting guides, and FAQs to empower researchers in systematically identifying and resolving these issues.
Troubleshooting Guides & FAQs
Q1: Why is there high inter-slide or inter-batch variability in staining intensity for the same target and sample type? A: This often points to pre-analytical or reagent variability. Follow this investigative protocol:
- Protocol: Reagent Stability & Aliquot Consistency Test
- Methodology: From a master batch of FFPE tissue sections (e.g., human tonsil), create three identical staining sets.
- Set A: Use a freshly prepared, aliquoted primary antibody dilution from a newly reconstituted vial.
- Set B: Use a primary antibody aliquot that has undergone 5 freeze-thaw cycles.
- Set C: Use a primary antibody dilution that was prepared 72 hours prior and stored at 4°C.
- Process all sets in the same automated stainer run. Quantify staining intensity (e.g., H-Score, DAB Optical Density) in identical anatomical regions across 10 fields per slide.
- Data & Analysis:
- Quantitative data from such an experiment typically shows significant degradation in Sets B and C.
| Test Set | Mean DAB OD (Target Region) | Coefficient of Variation (CV) Across Slides | Inferred Root Cause |
|---|---|---|---|
| Set A (Fresh Aliquot) | 0.45 | 8% | Baseline performance. |
| Set B (5 Freeze-Thaws) | 0.28 | 22% | Antibody degradation from improper storage. |
| Set C (72h Old Dilution) | 0.31 | 18% | Loss of antibody activity in diluted state. |
Q2: What could cause patchy or uneven staining across a single tissue section? A: This is frequently an artifact of the staining procedure itself. The primary suspect is inadequate or uneven reagent coverage during manual or automated steps.
- Protocol: Automated vs. Manual Dispense Validation
- Methodology: Select a serial section slide with known heterogeneous antigen distribution. Perform IHC using an automated platform with a calibrated, properly primed liquid dispensing system. On a duplicate slide, perform manual application of the same primary antibody, ensuring the reagent is applied but not spread evenly with a pipette tip. Compare staining patterns.
- Key Check: Inspect the automated stainer's liquid dispensing lines for bubbles and ensure the slide rack is level. For manual protocols, always use a hydrophobic barrier pen and cover the tissue with enough volume to form a convex meniscus.
Q3: How do I differentiate between true negative staining and a technical false negative? A: Implement a systematic set of controls within every run.
- Protocol: Comprehensive Control Slide Strategy
- Methodology: Include the following controls in each staining batch:
- Positive Control: A tissue known to express the target at a defined level.
- Negative Control: The same tissue type known to be null for the target (genetically confirmed if possible).
- Isotype Control: To assess non-specific binding of the primary antibody.
- Assay Positive Control (APC): A cell line pellet or tissue with known, stable expression of the target, processed identically to test samples. This controls for the entire assay process.
- Endogenous Enzyme Block Control: A section where the chromogen is applied after the block but without the primary antibody to check for incomplete peroxidase/alkaline phosphatase blockage.
- Methodology: Include the following controls in each staining batch:
Root Cause Analysis Framework: Decision Diagram
Title: Staining Inconsistency Root Cause Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Rationale for Robustness |
|---|---|
| Validated Primary Antibody with Lot-Specific Data Sheet | Core detection agent. Validation for IHC-specific applications (vs. WB) and consistent lot-to-lot performance data are critical for reproducibility. |
| Automated IHC Stainer & Certified Reagents | Minimizes operator-dependent variability. Use manufacturer-certified detection kits and buffers tailored for the platform. |
| Multitissue Microarray (TMA) Control Block | Contains defined positive, negative, and expression gradient tissues. Enables simultaneous monitoring of staining performance across dozens of samples on one slide. |
| Chromogen with Enhanced Stability (e.g., polymer-based DAB+) | Provides sharper signals and better resistance to solvent fading during coverslipping compared to traditional DAB. |
| Antigen Retrieval Buffer pH Standardization Kit | Precise pH (6.0 Citrate vs. 9.0 EDTA/Tris) is antigen-specific. Using a standardized, pH-verified buffer system prevents variable retrieval efficiency. |
| Protein Block (Species-Specific, Serum-Based) | Reduces non-specific background staining by blocking endogenous reactive sites on tissue, improving signal-to-noise ratio. |
| Barrier Pens (Hydrophobic) | Creates a defined, uniform incubation area for manual staining, preventing reagent spread and ensuring consistent volume-to-area ratio. |
| Digital Slide Scanner & Quantitative Image Analysis Software | Enables objective, high-throughput quantification of staining intensity and distribution, moving beyond subjective scoring. |
Troubleshooting Antigen Retrieval and Antibody Performance Across Platforms
This technical support center is designed to support researchers in achieving robust immunohistochemistry (IHC) assays, a critical requirement for multi-site reproducibility studies in drug development. Consistent antigen retrieval and antibody performance are foundational to reliable, comparable data across different laboratories and platforms.
Troubleshooting Guides & FAQs
Antigen Retrieval Issues
Q1: Why is my IHC staining weak or absent even with a validated antibody? A: This is frequently due to suboptimal antigen retrieval (AR). The fixation process cross-links and masks epitopes; AR reverses this. Troubleshoot using the following steps:
- Verify Fixation: Over-fixation (e.g., >72h in formalin) requires more aggressive AR. Under-fixation leads to poor morphology.
- Choose AR Method: Heat-Induced Epitope Retrieval (HIER) using a pressure cooker, microwave, or water bath is standard for most formalin-fixed paraffin-embedded (FFPE) samples. Proteolytic-Induced Epitope Retrieval (PIER) is suitable for some labile antigens.
- Optimize Buffer pH: Test a range (pH 6.0 Citrate vs. pH 8.0-9.0 Tris-EDTA). A shift in pH can dramatically alter staining intensity for specific epitopes.
- Control Time/Temperature: Standardize retrieval time and ensure the buffer reaches and maintains the intended temperature (e.g., 95-100°C for HIER).
Protocol: Standardized HIER Optimization Protocol
- Cut 4-5 serial sections from the FFPE block of interest.
- Deparaffinize and rehydrate sections.
- Perform HIER in a pressure cooker for 5 minutes at full pressure using different buffers: Citrate (pH 6.0), Tris-EDTA (pH 9.0), and a commercial high-pH buffer.
- Cool slides for 20-30 minutes at room temperature in the buffer.
- Proceed with the same IHC protocol for all slides (consistent antibody dilution, detection system, visualization).
- Compare signal intensity and background.
Q2: How do I address high background or non-specific staining post-retrieval? A: Over-retrieval can expose non-target epitopes or damage tissue architecture.
- Solution: Reduce HIER time or temperature incrementally.
- Include Controls: Always run a no-primary antibody control and an isotype control.
- Block Thoroughly: Use serum or protein blocks from the same species as the secondary antibody. Consider adding a casein-based block for charged residues exposed by AR.
Antibody Performance Across Platforms
Q3: The same antibody clone works on one automated stainer but fails on another. Why? A: Platform differences in reagent delivery, incubation timing, temperature, and wash stringency are key culprits.
Table 1: Key Variable Comparison Across Common Automated IHC Platforms
| Variable | Ventana BenchMark | Leica BOND | Dako Omnis | Agilent Ark |
|---|---|---|---|---|
| Default AR Chemistry | CC1 (pH ~8.5) or CC2 (pH ~6.0) | ER1 (pH 6.0) or ER2 (pH 9.0) | High pH (9.0) or Low pH (6.0) | Varies by protocol |
| Typical AR Method | Heated, pressurized | Heated, pressurized | Heated, pressurized | Heated, pressurized |
| Incubation Temp. | 36-37°C (primary) | Ambient or 37°C | Ambient | Ambient or 37°C |
| Wash Buffer | Proprietary Reaction Buffer | Bond Wash | Dako Wash | Tris-based |
| Detection System | UltraView, OptiView | Refine Polymer | EnVision FLEX | EnVision FLEX |
Solution: Re-optimize from the AR step when transitioning antibodies. Do not assume protocol transferability. Perform a checkerboard titration of antibody dilution against different AR conditions on the new platform.
Q4: How should I validate an antibody for a multi-site study? A: Follow a rigorous, pre-defined Standard Operating Procedure (SOP).
- Define Tissues & Controls: Use a standardized tissue microarray (TMA) containing known positive, negative, and variable expression tissues.
- Lock the Protocol: Fix AR method, buffer, time, antibody clone, catalog #, lot #, dilution, incubation time/temp, and detection kit.
- Cross-Platform Test: Run the locked protocol on all participating platforms.
- Quantitative Assessment: Use digital pathology/image analysis to score staining intensity (H-score, % positivity) and compare across sites.
Protocol: Multi-Site Antibody Validation Workflow
- Central SOP Development: Develop detailed IHC protocol with tolerances (e.g., antibody dilution ±10%, AR time ±2 min).
- Reagent Harmonization: Distribute aliquots of the same antibody lot, detection kit, and AR buffer to all sites.
- Shared TMAs: Circulate the same set of validation TMA slides to each site.
- Staining Run: Each site performs IHC per the SOP on their local platform.
- Digital Slide Scan & Analysis: Sites upload whole slide images to a centralized server for blinded, quantitative analysis.
- Data Review & Acceptance: Define pass/fail criteria (e.g., coefficient of variation for H-score < 15% across sites).
General Troubleshooting
Q5: My positive control stains well, but my experimental tissue does not. What's wrong? A: This indicates the protocol works, but the target antigen in your experimental tissue may be differentially affected.
- Considerations: Differences in fixation delay, fixation time, tissue type (e.g., brain vs. liver), or pre-analytical variables. Re-optimize AR specifically for the challenging tissue.
Q6: How do I manage lot-to-lot antibody variability? A:
- Bulk Purchase: Purchase a sufficient quantity of the same lot for the entire study.
- Bridging Experiment: When a new lot is required, run a parallel staining experiment with the old and new lots on the same TMA under identical conditions. Compare quantitative results before implementing the new lot.
Visualizations
Title: Antigen Retrieval Troubleshooting Decision Tree
Title: Cross-Platform Antibody Performance Problem & Solution
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Robust IHC Standardization
| Item | Function & Importance for Reproducibility |
|---|---|
| Validated Primary Antibody | Clone-specific, application-tested for IHC on FFPE. Bulk lot purchase minimizes variability. |
| Standardized AR Buffers | Consistent, pH-verified citrate or Tris-EDTA buffers. Critical for epitope exposure. |
| Automated IHC Platform | Provides precise control over incubation times, temperatures, and wash cycles. Reduces user variability. |
| Multitissue Control Blocks (TMA) | Contain known positive/negative tissues. Essential for run-to-run and site-to-site validation. |
| Polymer-based Detection System | High sensitivity and low background. Superior to avidin-biotin systems. Use same kit across sites. |
| Digital Pathology Scanner | Enables quantitative analysis (H-score, % positivity) and remote peer review for objective comparison. |
| Reference FFPE Cell Pellets | Cultured cells with known antigen expression, fixed and processed uniformly. Ideal process controls. |
Optimizing Signal-to-Noise Ratio and Counterstaining for Uniformity
Technical Support Center: Troubleshooting Guides & FAQs
This technical support resource is designed to address common challenges in immunohistochemistry (IHC) that directly impact the reproducibility of assays across multiple research sites, a core requirement for robust biomarker validation in drug development.
FAQ Section: Common Issues & Solutions
Q1: My positive staining is weak and variable across slides, despite using the same protocol. What are the primary causes? A: This is often a signal-to-noise ratio (SNR) issue stemming from pre-analytical variables. Key factors are:
- Fixation Time & Delay: Inconsistent fixation (under/over-fixation) dramatically alters antigen availability. See Table 1.
- Antigen Retrieval (AR) Inconsistency: pH, temperature, and time of AR buffer must be strictly controlled.
- Primary Antibody Incubation: Temperature fluctuations during incubation lead to variable binding kinetics. Protocol (Standardization): For a target like HER2, implement a calibrated automated stainer. Use a validated retrieval buffer (e.g., citrate, pH 6.0) at 95°C for 40 minutes (± 2 min). Primary antibody incubation must be at 37°C in a thermostatically controlled chamber for 30 minutes.
Q2: The background is high and non-uniform, obscuring specific signal. How can I reduce it? A: High background noise is frequently due to non-specific binding or endogenous enzyme activity.
- Blocking: Ensure complete and consistent blocking. Use 2.5% normal horse serum in a protein-based blocker for 30 minutes at RT.
- Antibody Concentration: Titrate every new antibody lot. A concentration that is too high increases background.
- Wash Stringency: Increase wash buffer molarity (e.g., 0.05M Tris vs. 0.1M Tris) and add a mild detergent (0.05% Tween-20).
- Endogenous Peroxidase: For HRC systems, use a fresh, potent peroxidase block (3% H₂O₂ in methanol) for 15 minutes.
Q3: My counterstain (hematoxylin) intensity varies from light blue to overly dark purple, affecting digital image analysis uniformity. How do I standardize it? A: Hematoxylin variability arises from inconsistent differentiation and bluing steps. Protocol (Standardization):
- Stain: Gill's Hematoxylin II for 3 minutes.
- Rinse: Run in tap water for 1 minute.
- Differentiate: Dip slides in 0.5% acid alcohol (0.5% HCl in 70% ethanol) 3-5 times (standardize dips).
- Bluing: Immerse in 0.1% ammonium hydroxide (or Scott's Tap Water) for 1 minute until nuclei are crisp blue.
- Rinse: Deionized water for 1 minute. Monitor hematoxylin oxidation state and replace after staining every 40 slides.
Q4: What quantitative metrics can I use to benchmark SNR and staining uniformity across sites? A: Utilize digital pathology image analysis to derive objective, quantitative data. Key metrics are summarized in Table 1.
Table 1: Quantitative Metrics for IHC Assay Robustness
| Metric | Description | Target for Robustness | Measurement Tool |
|---|---|---|---|
| Signal Intensity (Positive) | Mean optical density of DAB stain in target region. | CV < 15% across sites/slides. | Image Analysis Software (e.g., QuPath, HALO). |
| Background Intensity | Mean optical density of DAB stain in negative tissue region. | Should be < 10% of positive signal intensity. | Image Analysis Software. |
| Signal-to-Noise Ratio (SNR) | (Positive Signal Intensity – Background Intensity) / SD of Background. | SNR > 5 is generally acceptable; >10 is optimal. | Calculated from intensity measurements. |
| Counterstain Intensity | Mean optical density of hematoxylin stain in nucleus. | CV < 20% across sites/slides. | Image Analysis Software. |
| Positive Pixel Ratio | Percentage of pixels in Region of Interest (ROI) above a positive intensity threshold. | CV < 20% across sites/slides. | Image Analysis Software. |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Optimizing SNR & Uniformity |
|---|---|
| Validated Primary Antibody Clone | Ensures specific, reproducible binding to the target epitope. Lot-to-lot consistency is critical. |
| Automated IHC Stainer | Removes manual variability in incubation times, temperatures, and reagent application. |
| pH-Stable Antigen Retrieval Buffer | Consistent pH (±0.1) is vital for uniform epitope recovery. Use commercial, QC'd buffers. |
| Polymer-based Detection System | Amplifies signal while minimizing background vs. traditional avidin-biotin (ABC) systems. |
| Stable DAB Chromogen | Single-component, metal-enhanced DAB provides consistent precipitate and higher SNR. |
| Standardized Hematoxylin | A ready-to-use, filtered hematoxylin with a defined oxidation state ensures counterstain uniformity. |
| Mounted Slide Scanner | Enables whole-slide imaging for quantitative, high-throughput analysis of staining uniformity. |
Experimental Protocols for Key Validations
Protocol 1: Primary Antibody Titration for Optimal SNR
- Prepare serial dilutions of primary antibody (e.g., 1:50, 1:100, 1:200, 1:500) on a multi-tissue block containing known positive and negative tissues.
- Run IHC using standardized AR, detection, and DAB steps.
- Scan slides and quantify signal intensity and background in defined ROIs.
- Calculate SNR for each dilution. The optimal dilution is the one just before the point where signal intensity plateaus but background continues to increase.
Protocol 2: Inter-Site Staining Reproducibility Test
- Central Preparation: A central lab prepares and distributes identical tissue microarray (TMA) slides from the same block lot, pre-cut and stored under inert gas.
- Distributed Staining: Participating sites stain slides using the identical, written protocol, equipment logs, and specified reagent lots.
- Central Analysis: All slides are returned to the central lab for scanning with a calibrated scanner.
- Data Collection: A single analyst measures the metrics in Table 1 across all TMA cores from all sites.
- Statistical Analysis: Calculate the intra-class correlation coefficient (ICC) and coefficients of variation (CV) for each metric. An ICC > 0.9 indicates excellent inter-site reproducibility.
Visualization: Critical Pathways and Workflows
Title: Key Factors Influencing IHC Signal-to-Noise Ratio
Title: Standardized Workflow for Multi-Site IHC Reproducibility
Calibration and Maintenance Schedules for Critical Equipment (Stainers, Scanners).
This technical support center is established to support multi-site reproducibility research in IHC, where assay robustness is fundamentally dependent on the consistent performance of automated stainers and digital slide scanners. Standardized calibration and maintenance are non-negotiable prerequisites.
Troubleshooting Guides & FAQs
Q1: After routine maintenance on our autostainer, we observe uneven DAB chromogen deposition across the slide. What is the likely cause and how can we resolve it? A: This is typically caused by air bubbles trapped within the fluidic lines or dispenser nozzles post-maintenance. Follow this protocol:
- Run the stainer's prime/purge utility for all reagent lines, especially the chromogen line.
- Visually inspect all lines for persistent bubbles.
- Execute a diagnostic run using a water-soluble dye on a blank slide to assess dispensing uniformity.
- If unevenness persists, manually clean the specific dispenser nozzle with recommended buffer using a laboratory wipe.
Q2: Our digital scanner is producing images with inconsistent focus, manifesting as localized blurring in specific tissue regions (e.g., dense lymphoid areas). How should we address this? A: Inconsistent focus often indicates a need for calibration of the autofocus system or contamination of the optical path.
- Optical Path Check: Power down the scanner. Use an air duster and approved optical wipes to gently clean the exterior of the objective lenses and the glass scanning stage.
- Autofocus Calibration: Run the manufacturer's proprietary "Autofocus Calibration" slide protocol. This slide has a known topographical profile to recalibrate the Z-axis motor and sensor.
- Tissue Detection Settings: Review the scan profile. For heterogeneous tissues, enable "multipoint" or "tissue map" focusing rather than single-point focus.
Q3: We are noting a drift in H-Score values for our same control tissue scanned monthly over 6 months. The stainer protocol is unchanged. Could the scanner be implicated? A: Yes. Scanner lamp intensity degrades over time, leading to decreased signal intensity and altered quantitative results.
- Lamp Hours Audit: Check the logged usage hours of the scanner's halogen or LED light source against its rated lifespan (typically 1000-2000 hours).
- Calibration Verification: Scan a NIST-traceable photometric calibration slide (e.g., a slide with known optical density patches).
- Analysis: Extract mean pixel intensity values for each patch. Compare to baseline values from when the lamp was new. A deviation >10% indicates a need for lamp replacement and full optical calibration.
The following tables synthesize manufacturer specifications and consensus guidelines from recent laboratory medicine literature to ensure equipment performance aligns with reproducibility study demands.
Table 1: Recommended Calibration Schedule for IHC Critical Equipment
| Equipment | Calibration Task | Frequency | Performance Tolerance | Purpose in Reproducibility |
|---|---|---|---|---|
| Autostainer | Dispense Volume Verification | Quarterly | ±5% of set volume (e.g., 100µL ±5µL) | Ensures consistent reagent delivery and antigen-antibody reaction kinetics. |
| Autostainer | Temperature Verification (Bake, Dewax, Retrieval) | Monthly | ±2°C of set point | Critical for consistent epitope retrieval and enzyme-mediated detection. |
| Digital Scanner | Photometric Calibration | Monthly | ±5% intensity uniformity across field | Ensures quantitative image data stability for longitudinal studies. |
| Digital Scanner | Spatial Calibration | Annual | ±2% dimensional accuracy (µm/pixel) | Guarantees accurate morphometric measurements across sites. |
| Both | Daily/Pre-Run Check | Control slide (positive/negative) | Consistent expected staining pattern | Operational verification of the entire IHC workflow. |
Table 2: Preventive Maintenance Schedule & Key Actions
| Equipment | Maintenance Task | Frequency | Key Materials & Actions | Impact on Assay Robustness |
|---|---|---|---|---|
| Autostainer | Fluidic Path Deep Clean | Monthly | 10% Bleach, 70% Ethanol, DI Water. Run purge protocols. | Prevents reagent carryover and biofilm formation that cause batch effects. |
| Autostainer | Reagent Probe Wiping & Alignment Check | Weekly | Lint-free wipes, alignment jig. | Prevents droplet formation, ensures accurate dispensing onto tissue. |
| Digital Scanner | Mechanical Stage & Rail Cleaning | Weekly | Dry, lint-free cloth. | Prevents slide positioning errors and focus failures. |
| Digital Scanner | Optical Component Cleaning | Quarterly (or as needed) | Compressed air, approved optical wipes. | Maintains image sharpness and color fidelity. |
| Digital Scanner | Light Source Replacement | As per hours log (≈1500 hrs) | OEM Halogen/LED lamp. | Prevents signal drift in quantitative image analysis (QIA). |
Experimental Protocol: Monthly Photometric Calibration of a Digital Scanner
Objective: To verify and correct for any drift in the scanner's optical detection system, ensuring stable digital signal output for quantitative IHC. Materials: NIST-traceable reflectance calibration slide, scanner software. Methodology:
- Baseline Establishment (Day 0): On a new or freshly calibrated lamp, scan the calibration slide using a "Calibration Scan" profile. The software automatically records the mean pixel intensity (e.g., 0-255 scale) for each pre-defined patch of known reflectance.
- Monthly Verification:
- Perform the same "Calibration Scan" under identical software settings.
- The software compares current patch intensities to the baseline.
- It generates a correction matrix (flat-field) if deviations are within correctable limits (typically <15% drift).
- This matrix is automatically applied to all subsequent research scans.
- Failure Action: If deviation exceeds the correctable limit, the software will flag the need for lamp replacement. After replacement, a new Day 0 baseline must be established.
The Scientist's Toolkit: Research Reagent Solutions for IHC Reproducibility
| Item | Function in IHC Assay Robustness |
|---|---|
| Validated Primary Antibody Clone | Identical clone and lot sourcing across sites is critical to control for epitope specificity and affinity variability. |
| Automated Stainer-Compatible Detection Kit | Standardized polymer-based detection systems minimize variability compared to manually prepared ABC methods. |
| NIST-Traceable Calibration Slide | Provides an objective, standardized target for scanner photometric and spatial calibration across instruments and sites. |
| Multi-Tissue Control Block | Contains cell lines or tissues with known, graded expression of targets. Run on every slide/batch for process monitoring. |
| Bench-Stable, Ready-to-Use Buffers | Pre-mixed, pH-stable retrieval buffers and wash buffers eliminate a major source of inter-operator and inter-site preparation variance. |
Diagrams
Diagram 1: IHC Reproducibility Workflow with QC Nodes
Diagram 2: Scanner Signal Drift Impact on Quantitative IHC
The Role of Reference Standards and Control Tissues in Continuous Monitoring
Within multi-site reproducibility research for IHC assays, continuous monitoring is critical for ensuring longitudinal data integrity. Reference standards and control tissues form the backbone of this process, allowing for the calibration of instruments, validation of protocols, and normalization of results across time and locations.
Troubleshooting Guides & FAQs
Q1: Our positive control tissue shows weak or inconsistent staining over time, despite using the same protocol. What are the primary causes? A: This is a common issue in continuous monitoring. Primary causes include:
- Degradation of the Control Tissue Block: Even properly stored FFPE blocks can undergo antigen degradation over years.
- Variability in Antigen Retrieval: Subtle differences in retrieval solution pH, temperature, or time significantly impact results.
- Lot-to-Lot Variation in Primary Antibodies: Different antibody clones or production lots have varying affinities.
- Slide Storage Conditions: Stained or unstained control slides stored for monitoring may degrade if not sealed and stored in a -20°C freezer.
Q2: How do we select the appropriate reference standard for a new biomarker assay intended for multi-site use? A: Follow this hierarchy:
- Commercial Certified Reference Materials (CRMs): Prioritize these if available for your target (e.g., from NIST or IVD manufacturers).
- Characterized Cell Line Microarrays (CCLMs): Use cell lines with known, stable expression levels of the target, formalin-fixed and embedded in a microarray format.
- In-House Reference Tissues: If commercial options are unavailable, create a large, homogeneous master block from a well-characterized tissue sample. Validate it extensively before distribution.
Q3: What is the minimum set of controls required for each staining run in a longitudinal study? A: Each run should include:
- Positive Tissue Control: A tissue with known, moderate expression of the target.
- Negative Tissue Control: A tissue known to lack the target antigen.
- Reagent Negative Control (e.g., IgG control): To identify non-specific antibody binding.
- Background/Retrieval Control: A section stained omitting the primary antibody to assess detection system noise.
Data Presentation: Key Metrics for Monitoring
Table 1: Quantitative Metrics for Continuous Monitoring of IHC Assay Performance
| Performance Indicator | Target Value | Acceptable Range | Measurement Frequency | Corrective Action Trigger |
|---|---|---|---|---|
| Positive Control Staining Intensity (Score) | Consistent with historical median | Median ± 1.0 units (e.g., H-Score) | Every run | Deviation >1.5 units |
| Negative Control Staining | 0 (No specific staining) | Score of 0-1 | Every run | Any specific staining > Score 1 |
| Inter-Site Coefficient of Variation (CV) | < 15% | < 20% | Quarterly | CV > 20% for 2 consecutive periods |
| Reference Standard Quantification (e.g., H-score, % positivity) | Established baseline | Baseline ± 2 Standard Deviations | Per run for slide; quarterly for block | Point outside 3SD limits |
Experimental Protocols
Protocol 1: Establishing an In-House Longitudinal Control Tissue Block
- Source Selection: Identify a surgical tissue specimen with homogeneous, moderate expression of the target antigen. Obtain necessary IRB/ethical approvals.
- Processing: Fix the entire tissue in 10% Neutral Buffered Formalin for 18-24 hours at room temperature. Process and embed in a single, large paraffin block using a standardized protocol.
- Validation: Cut 50 serial sections. Every 5th section is stained with the target IHC assay alongside a validated commercial control (if available). Scores (e.g., H-score, % positivity) are recorded by three independent, blinded pathologists.
- Characterization: Calculate the mean and standard deviation of the scores. This block becomes the "golden master." Store at 4°C in a sealed container with desiccant.
- Distribution: For multi-site studies, create identical daughter blocks from the master or distribute pre-cut, unstained slides from a single large-sectioning event, stored at -20°C.
Protocol 2: Quarterly Performance Review for Multi-Site Reproducibility
- Central Distribution: The coordinating center sends pre-cut sections from the same reference standard block to all participating sites.
- Parallel Staining: All sites stain their slides within a 2-week window using the shared, detailed SOP (including specified antibody lot, retrieval method, and detection kit).
- Digital Scan & Analysis: Sites upload whole-slide images to a centralized platform. A single analyst scores all images using a pre-defined, validated digital image analysis algorithm.
- Data Collation & Analysis: Calculate the inter-site CV for the quantification data (e.g., H-score). Generate a Levey-Jennings chart to visualize each site's result against the established mean and control limits.
- Corrective Action: Sites whose results fall outside 3SD are required to perform root-cause analysis (e.g., reagent calibration, instrument maintenance) and repeat the test.
Mandatory Visualizations
Title: Multi-Site IHC Monitoring & Corrective Action Workflow
Title: How Reference Standards Ensure IHC Assay Robustness
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for IHC Continuous Monitoring
| Item | Function | Key Consideration for Reproducibility |
|---|---|---|
| Certified Reference Material (CRM) | Provides an analyte-specific, traceable standard for calibration and validation. | Ensure it is commutable with clinical patient samples. |
| Cell Line Microarray (CMA) Block | Contains multiple cell lines with defined expression levels, offering internal controls on one slide. | Ideal for monitoring antibody specificity and detection system linearity. |
| "Golden Master" Tissue Block | A large, homogeneous in-house control tissue block for longitudinal consistency. | Characterize thoroughly before use; limit number of sections from a single block. |
| Multiplex IHC/IF Control Tissues | Tissues with known co-expression patterns of multiple targets. | Essential for validating multiplex assay panels and spectral unmixing. |
| Digital Image Analysis Software | Quantifies staining intensity and distribution objectively. | Use the same algorithm and version across all sites in a study. |
| Slide Staining Logbook (Digital) | Tracks reagent lots, incubation times, instrument IDs, and operators for every run. | Critical for root-cause analysis during performance drift. |
Proving Your Assay Works: Validation Strategies and Tool Comparison
Troubleshooting Guides & FAQs
Q1: Our IHC staining shows high background, making specific signal quantification unreliable. How does this impact the calculation of Sensitivity and Specificity? A1: High background increases false positives, directly reducing assay specificity. To troubleshoot:
- Perform an antibody titration to find the optimal dilution that minimizes non-specific binding.
- Include a negative control without primary antibody to identify background level.
- Increase the stringency of washes (e.g., adjust salt concentration, add mild detergent).
- Use a blocking serum from the same species as the secondary antibody.
Q2: When validating an IHC assay across multiple laboratory sites, we see high inter-observer variability in scoring. How does this affect Precision metrics? A2: Low inter-rater agreement (e.g., low Cohen's Kappa score) indicates poor precision/reproducibility, a critical failure for multi-site studies.
- Solution: Implement digital image analysis (DIA) using standardized algorithms. Provide extensive training with a consensus scoring guide featuring representative images for each score. Regular reconciliation meetings are essential.
Q3: Our positive control tissue shows weak staining, suggesting low assay Sensitivity. What are the key steps to investigate? A3:
- Check Antigen Retrieval: Optimize method (heat-induced vs. enzymatic), pH of buffer (6.0 vs. 9.0), and retrieval time.
- Primary Antibody: Verify antibody specificity (e.g., knockout tissue validation), check concentration, and ensure proper storage (no repeated freeze-thaw).
- Detection System: Ensure the detection kit is not expired. Amplification steps may be needed for low-abundance targets.
Q4: How do we accurately determine "True Negatives" for calculating Specificity in IHC? A4: True Negatives require confirmed negative samples. Best practices include:
- Using tissue microarrays (TMAs) containing validated negative tissues (e.g., tissues known not to express the target via other methods like RNA-seq).
- Including isotype controls for monoclonal antibodies.
- Using genetic knockout or knockdown tissue sections as the gold standard negative control.
Table 1: Core Definitions of IHC Validation Metrics
| Metric | Formula (Conceptual) | Interpretation in IHC Context |
|---|---|---|
| Accuracy | (TP + TN) / (TP+TN+FP+FN) | Overall agreement of IHC result with a gold standard truth. |
| Precision (Repeatability/Reproducibility) | e.g., ICC, Cohen's Kappa | Consistency of results upon repeat testing (intra-site) or across sites/observers (inter-site). |
| Sensitivity (True Positive Rate) | TP / (TP + FN) | Ability of the assay to correctly identify antigen-positive cells/tissues. |
| Specificity (True Negative Rate) | TN / (TN + FP) | Ability of the assay to correctly exclude antigen-negative cells/tissues. |
Table 2: Example Data from a Multi-Site IHC Validation Study
| Site | Sensitivity (%) | Specificity (%) | Inter-Observer Agreement (Kappa) | Intra-Site Precision (ICC) |
|---|---|---|---|---|
| Site A | 95 | 88 | 0.75 | 0.92 |
| Site B | 89 | 92 | 0.81 | 0.94 |
| Site C | 97 | 85 | 0.68 | 0.89 |
| Consensus Target | ≥90 | ≥85 | ≥0.70 | ≥0.90 |
Detailed Experimental Protocol: IHC Assay Validation for Multi-Site Reproducibility
Title: Standardized Protocol for Multi-Site IHC Validation of Biomarker X.
Objective: To establish a robust, reproducible IHC assay for Biomarker X across three independent laboratories.
Materials: See "The Scientist's Toolkit" below.
Methods:
- Sample Set: A TMA containing 60 cores is created, comprising:
- 20 known positive cores (confirmed by Western blot).
- 20 known negative cores (confirmed by RNA-seq).
- 10 cores with variable expression levels.
- 10 cores for staining controls.
- Reagent Standardization: All sites use the same lot of primary antibody, detection kit, and chromogen. Buffer recipes are standardized.
- Staining Procedure:
- Deparaffinization & Rehydration: Standard xylene/ethanol series.
- Antigen Retrieval: Heat-Induced Epitope Retrieval (HIER) in pH 9.0 EDTA buffer for 20 minutes in a pressurized decloaking chamber.
- Peroxidase Blocking: 3% H₂O₂ for 10 minutes.
- Protein Block: 2.5% normal horse serum for 20 minutes.
- Primary Antibody: Incubate with anti-Biomarker X (1:200 dilution) for 60 minutes at room temperature.
- Detection: Apply polymer-based HRP-conjugated secondary antibody for 30 minutes, followed by DAB chromogen for 5 minutes.
- Counterstain: Hematoxylin for 1 minute.
- Digital Image Analysis (DIA): All stained slides are scanned at 20x magnification. A pre-defined algorithm quantifies the percentage of DAB-positive nuclei (H-score) in a blinded manner.
- Statistical Analysis:
- Sensitivity/Specificity: Calculate against the pre-defined truth using DIA H-score > 15 as positive.
- Precision (Inter-site): Calculate Intraclass Correlation Coefficient (ICC) for H-scores from all sites.
- Inter-observer Agreement: Three pathologists score a subset of 30 images; calculate Fleiss' Kappa.
Diagrams
Title: IHC Validation Workflow for Multi-Site Studies
Title: Relationship of IHC Validation Metrics
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Robust IHC Validation
| Item | Function in Validation | Example/Note |
|---|---|---|
| Validated Primary Antibody | Binds specifically to the target antigen. Critical for sensitivity and specificity. | Use CRISPR-knockout validated antibody; specify clone, host species, and catalog number. |
| Polymer-based Detection System | Amplifies signal from primary antibody with high sensitivity and low background. | Reduces non-specific staining vs. traditional avidin-biotin systems. |
| Reference Tissue Microarray (TMA) | Contains pre-characterized positive, negative, and variable tissues. Essential for determining true positives/negatives. | Commercial or custom-built. Core size and number should be standardized. |
| Automated Staining Platform | Ensures consistent timing, temperature, and reagent application across runs and sites. | Key for achieving high precision (repeatability). |
| Digital Slide Scanner & Analysis Software | Enables objective, quantitative scoring (H-score, % positivity). Eliminates observer bias for precision metrics. | Scan resolution and analysis algorithm parameters must be identical across sites. |
| Standardized Buffer Kits | Provides consistent antigen retrieval and washing conditions. | Pre-mixed, pH-verified buffers prevent inter-site variability. |
| Control Slides | Monitor technique performance. Includes: 1) Positive tissue control, 2) Negative reagent control, 3) Isotype control. | Must be included in every staining run. |
Comparative Analysis of Commercial vs. Laboratory-Developed Controls
Within multi-site IHC assay reproducibility research, the choice between commercial and laboratory-developed controls (LDCs) is a critical variable influencing data robustness. This technical support center addresses common experimental issues, framed within a thesis on optimizing control strategies to minimize inter-site variability in drug development research.
Troubleshooting Guides & FAQs
Q1: We observe high inter-site staining intensity variance using a commercial control. What are the primary troubleshooting steps? A: This often relates to pre-analytical variables. First, verify that all sites are using the same lot of the commercial control and have validated the antigen stability under their specific fixation and storage protocols. Ensure standardized slide cutting thickness (typically 4-5 µm) across sites. Perform a calibration run using a shared instrument service protocol to rule out autostainer variability. If variance persists, consider if the commercial control's antigen expression level is at an extreme high or low end, making subtle technical differences more pronounced.
Q2: Our laboratory-developed control shows degradation over time. How can we improve its shelf-life? A: LDC stability is paramount. Implement a rigorous validation protocol:
- Fixation Audit: Standardize neutral buffered formalin fixation time precisely (e.g., 24-48 hours).
- Processing Consistency: Use a dedicated tissue processor for control tissues only.
- Storage: Aliquot control blocks and store at -20°C in vacuum-sealed bags with desiccant. For cut sections, use coated slides and store at -80°C under nitrogen atmosphere.
- Stability Monitoring: Establish a schedule to re-stain a reference slide from the master block every 6 months using a validated commercial control as a comparator to track signal decay.
Q3: When validating a new LDC, how do we establish its acceptable expression range? A: Develop a statistical baseline through a "round-robin" protocol.
- Cut 30 sequential sections from the candidate LDC block.
- Stain 10 sections at three different sites using the identical, calibrated IHC protocol.
- Perform quantitative image analysis (e.g., H-score, % positive cells) using a shared digital pathology algorithm.
- Calculate the mean expression and standard deviation (SD). The acceptable range for future runs is typically set at mean ± 2SD. This range must be tighter than the clinical or biological cut-off you are studying.
Q4: A commercial control batch has changed, causing a shift in our assay's scoring. How should we respond? A: Do not bridge data directly. Initiate a formal re-validation:
- Stain the old and new control lots alongside a well-characterized internal LDC or archived patient sample.
- Run a full standard curve if using semi-quantitative methods.
- Document the delta in staining intensity and patterns. Update the assay's scoring guidelines or positivity thresholds based on the new control. All data generated with the previous lot must be analyzed separately.
Table 1: Key Attribute Comparison of Control Types
| Attribute | Commercial Controls | Laboratory-Developed Controls |
|---|---|---|
| Lot-to-Lot Consistency | High (Manufacturer QC) | Variable (In-house dependent) |
| Initial Cost | High per unit | Low (once established) |
| Long-Term Cost | Recurring expense | Primarily labor/maintenance |
| Customization | Low (Fixed antigens/levels) | High (Tissue type, expression level) |
| Availability | Immediate, but supply-chain risk | Continuous, self-reliant |
| Documentation | Certificate of Analysis (CoA) | Internally generated SOPs & records |
| Multi-Site Standardization | Facilitates through identical source | Requires rigorous shared validation |
Table 2: Impact of Control Choice on Multi-Site CV% (Coefficient of Variation)
| Control Strategy | Average Inter-Site CV% (Staining Intensity) | Key Contributing Factor |
|---|---|---|
| Validated Commercial Control | 8-15% | Instrument/platform variability |
| Un-Validated LDC | 20-35% | Pre-analytical and block heterogeneity |
| Centrally-Validated & Distributed LDC | 10-18% | Section-to-section variability |
| Digital Image Analysis with Reference Control | 5-12% | Algorithm and ROI selection |
Experimental Protocols
Protocol 1: Validation of a Laboratory-Developed Control for Multi-Site Use
Objective: To create and validate an LDC that ensures ≤15% inter-site CV for a specific IHC assay. Materials: See "The Scientist's Toolkit" below. Methodology:
- Tissue Selection: Identify residual surgical pathology tissue with moderate, homogeneous expression of the target antigen. Obtain necessary IRB/ethical approval.
- Standardized Processing: Process the tissue in a single, continuous run in neutral buffered formalin for 24 hours. Use a standardized dehydration and paraffin-embedding protocol.
- Master Block Creation: Create a large "master" block via a tissue microarrayer or by embedding a large section in a single block. Perform initial staining on 10 sections from different block depths to confirm homogeneity.
- Pre-Cutting for Distribution: Cut 500-1000 sections at 4 µm. Float on ionized water, mount on positively charged slides. Air dry overnight.
- Baking & Storage: Bake slides at 60°C for 1 hour. Place in slide boxes with desiccant, purge with nitrogen, and seal. Store at -80°C.
- Multi-Site Validation: Distribute 30 slides each to three participating sites. Each site stains 10 slides over three different days using a shared, detailed SOP.
- Analysis & Acceptance Criteria: Perform quantitative analysis. The LDC is validated if the inter-site CV is ≤15% and the intra-site CV is ≤10%.
Protocol 2: Parallel Testing for Control Comparability
Objective: To compare the performance of a new commercial control lot against an expiring lot or an LDC. Methodology:
- Slide Preparation: Label slides for both controls and include a known patient sample.
- Staining Run: Place all controls on the same run using the standard IHC protocol. Repeat on three separate runs.
- Image Acquisition: Scan slides using a standardized 20x objective and fixed exposure settings.
- Quantification: Use the same digital analysis template to measure staining intensity (e.g., DAB optical density) and percentage of positive cells in predefined regions.
- Statistical Analysis: Perform a Student's t-test or ANOVA. A non-significant difference (p > 0.05) in intensity and % positivity indicates comparability. Document any observable pattern differences.
Visualizations
Diagram 1: IHC Control Validation Workflow
Diagram 2: Factors Affecting Multi-Site IHC Reproducibility
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in IHC Control Context |
|---|---|
| Neutral Buffered Formalin (10%) | Standardized fixative to preserve tissue architecture and antigenicity uniformly. |
| Paraffin Embedding Medium | Provides stable, consistent support for tissue sectioning. Low-melt point varieties aid homogeneity. |
| Positively Charged Microscope Slides | Ensures maximal adhesion of tissue sections during IHC staining protocols, preventing wash-off. |
| Desiccant Packs & Nitrogen Gas | Critical for long-term, degradation-free storage of cut control slides by removing oxygen and moisture. |
| Reference Standard (e.g., CRM) | Commercial reference material with independently verified values, used to calibrate assays and validate LDCs. |
| Digital Slide Scanner | Enables high-throughput, standardized image capture for quantitative analysis across multiple sites. |
| Quantitative Image Analysis Software | Allows objective measurement of staining intensity (DAB OD) and percentage positivity, reducing scorer bias. |
| Bar-Coded Slide Labeling System | Tracks control slides for lot, cut date, and staining run, ensuring traceability in multi-site studies. |
Statistical Methods for Assessing Inter-Site and Inter-Observer Concordance
Troubleshooting Guides & FAQs
FAQ 1: Why is our Fleiss' Kappa value unexpectedly low despite high percent agreement in our multi-site IHC scoring study?
- Answer: Percent agreement does not account for agreement occurring by chance. A high percent agreement with a low Kappa suggests that the prevalence of a particular score (e.g., most samples are "low") is high, making agreement by chance likely. Kappa corrects for this chance agreement. Review your score distribution. If it's highly skewed, consider using Prevalence-Adjusted Bias-Adjusted Kappa (PABAK) or reporting both metrics.
FAQ 2: During Inter-Class Correlation (ICC) analysis for continuous IHC data (e.g., H-Score), which model (one-way random, two-way random, or two-way mixed) should we select?
- Answer: The model depends on your experimental design.
- Use a Two-Way Random-Effects Model (ICC(2,1)) if both your sites/observers and your samples are considered randomly selected from larger populations, and you intend to generalize the reliability results. This is most common for multi-site reproducibility studies.
- Use a Two-Way Mixed-Effects Model (ICC(3,1)) if your sites/observers are the only ones of interest (a fixed set) and you do not wish to generalize beyond them.
- Use a One-Way Random-Effects Model (ICC(1,1)) if you are only accounting for sample variance and not systematically modeling site/observer effects.
FAQ 3: We encountered missing data for some samples at certain sites in our concordance study. How should we handle this for statistical analysis?
- Answer: Do not use simple mean imputation, as it can artificially inflate concordance. Preferred methods include:
- Complete Case Analysis: Use only samples with data from all sites/observers. This is valid only if data is Missing Completely at Random (MCAR) and doesn't severely reduce sample size.
- Multiple Imputation: Create several plausible values for the missing data based on other observed variables, analyze each dataset, and pool results. This is robust but complex.
- Use Concordance Metrics Tolerant of Missing Data: Some implementations of ICC and Cohen's Kappa for multiple raters can handle balanced missing data. Always report the amount and pattern of missingness.
FAQ 4: What is the minimum number of samples and observers required for a robust inter-observer concordance study for a new IHC assay?
- Answer: There is no universal minimum, but general guidelines exist based on statistical power. For a preliminary study, aim for:
- Samples: At least 30-50 samples covering the full range of expected expression (negative, weak, moderate, strong).
- Observers/Readers: At least 3-5, preferably from different sites to capture inter-site variability.
- Replicates: Each observer should score each sample at least twice in a blinded, randomized order to assess intra-observer reproducibility. A power analysis based on a pilot study is the gold standard for determining final sample size.
FAQ 5: How do we interpret a "moderate" ICC value? Is it sufficient for assay validation?
- Answer: "Moderate" is a qualitative label. Interpretation must be context-dependent.
- Benchmarks: Common benchmarks are: <0.5 Poor; 0.5-0.75 Moderate; 0.75-0.9 Good; >0.9 Excellent.
- Context is Key: For a critical diagnostic biomarker, you may require ICC > 0.9. For exploratory research, ICC > 0.7 might be acceptable. Always report the confidence interval for the ICC. An ICC of 0.7 with a 95% CI of 0.5 to 0.85 indicates substantial uncertainty. The pre-defined acceptable lower bound in your study protocol is ultimately what determines sufficiency.
Table 1: Common Concordance Statistics & Their Application in IHC
| Statistic | Data Type | Use Case | Interpretation Range | Key Consideration |
|---|---|---|---|---|
| Intraclass Correlation Coefficient (ICC) | Continuous (H-Score, % positive cells) | Assessing reliability of measurements across multiple sites or observers. | 0 to 1 (1 = perfect reliability) | Choose the correct model (one-way, two-way, random/mixed). |
| Cohen's Kappa (κ) | Binary (Positive/Negative) | Agreement between two observers on a categorical call. | -1 to 1 (1 = perfect agreement) | Affected by prevalence of the positive call. |
| Fleiss' Kappa (κ) | Ordinal or Binary (e.g., 0,1+,2+,3+) | Agreement among more than two observers on a categorical scale. | -1 to 1 (1 = perfect agreement) | Also sensitive to prevalence and number of categories. |
| Prevalence-Adjusted Bias-Adjusted Kappa (PABAK) | Binary | Agreement when prevalence is very high or low, or when there is systematic bias. | -1 to 1 (1 = perfect agreement) | Corrects for extreme prevalence which can distort Kappa. |
| Concordance Correlation Coefficient (CCC) | Continuous | Measuring agreement between two measurement methods (e.g., digital vs. manual scoring). | 0 to 1 (1 = perfect agreement) | Measures both precision (Pearson's r) and accuracy (bias from 45° line). |
Table 2: Example Concordance Results from a Multi-Site IHC Study
| Biomarker | Scoring Method | Sites (n) | Samples (n) | Primary Metric | Result (95% CI) | Interpretation |
|---|---|---|---|---|---|---|
| PD-L1 (SP263) | Tumor Proportion Score (TPS) % | 5 | 50 | ICC (Two-Way Random) | 0.87 (0.81, 0.92) | Good inter-site reliability |
| ER (SP1) | H-Score (0-300) | 3 | 40 | ICC (Two-Way Random) | 0.92 (0.88, 0.95) | Excellent inter-site reliability |
| HER2 (4B5) | Binary (Positive/Negative) | 6 | 100 | Fleiss' Kappa | 0.62 (0.55, 0.69) | Moderate inter-observer agreement |
| Ki-67 (30-9) | % Positive Nuclei | 4 | 60 | CCC (vs. Reference Lab) | 0.79 (0.72, 0.84) | Moderate to good agreement with reference |
Experimental Protocols
Protocol 1: Core Protocol for a Multi-Site Inter-Site Concordance Study
- Assay Standardization: All participating sites perform initial training using the same protocol, validated antibody clone, staining platform, and scanner model (if digital). A centrally stained control tissue microarray (TMA) is circulated to confirm initial staining concordance.
- Sample Selection & Distribution: A cohort of formalin-fixed, paraffin-embedded (FFPE) tissue samples (N ≥ 30), representing the full dynamic range of expression and relevant tumor types, is selected. Identical tissue sections are distributed to each site.
- Blinded Staining & Scanning: Each site stains the sample set in a single batch following the standardized protocol. Slides are digitized using a pre-defined scanner and settings (e.g., 20x magnification, consistent file format).
- Centralized, Blinded Analysis: All digital slides are uploaded to a central server. Analysis is performed by either:
- Pathologist Scoring: At least three certified pathologists, blinded to site and each other's scores, assess all slides using a defined scoring algorithm (e.g., H-Score, TPS).
- Digital Algorithm: A single validated image analysis algorithm is applied to all slides.
- Statistical Analysis: For continuous scores (H-Score), calculate ICC using a Two-Way Random-Effects Model to assess site-to-site variability. For categorical scores, calculate Fleiss' Kappa. Report point estimates with 95% confidence intervals.
Protocol 2: Inter-Observer Reproducibility Assessment for a New Scoring Guideline
- Observer Training: Develop a detailed scoring manual with representative images for each score category. Conduct a centralized training session for all observers (e.g., 5 pathologists).
- Test Set Creation: Assemble a test set of 40-60 digital slides that evenly represent all score categories and borderline cases.
- Scoring Rounds: Observers score the test set in two separate rounds, spaced at least 4 weeks apart. The slide order is randomized differently in each round.
- Data Collection: Scores are collected electronically.
- Statistical Analysis:
- Intra-Observer Concordance: Calculate Weighted Kappa (for ordinal scores) or ICC for each observer between Round 1 and Round 2 scores.
- Inter-Observer Concordance: Calculate Fleiss' Kappa (for ordinal) or ICC (for continuous) using the scores from the first round (or the average of both rounds) across all observers.
Diagrams
Diagram 1: Multi-Site IHC Concordance Study Workflow
Diagram 2: Statistical Model Selection for ICC
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for IHC Concordance Studies
| Item | Function & Importance for Concordance |
|---|---|
| Validated Primary Antibody Clone | The specific clone (e.g., PD-L1 clone 22C3) is critical. Different clones can have different binding affinities and epitopes, causing major inter-site variability. Use the same clone across all sites. |
| ISO 9001-Certified Detection Kit | A polymer-based detection system (e.g., EnVision, UltraView) ensures consistent amplification of the signal. Lot-to-lot consistency from a certified manufacturer minimizes variability. |
| Reference Control Cell Lines/Tissues | Commercially available FFPE cell pellets or tissue microarrays with known expression levels (negative, low, high) are essential for daily run validation and monitoring staining drift at each site. |
| Automated Staining Platform | Using the same model of automated stainer (e.g., Ventana Benchmark, Leica Bond) across sites standardizes incubation times, temperatures, and wash steps, greatly enhancing reproducibility. |
| Whole Slide Scanner & Software | For digital analysis, identical scanner models and software versions ensure consistent image quality, resolution, and color calibration, allowing valid comparisons of digital scores. |
| Digital Image Analysis (DIA) Software | A validated, scripted DIA algorithm (e.g., QuPath, HALO, Visiopharm script) removes observer subjectivity for continuous measures (e.g., H-Score, cell count), improving inter-observer ICC. |
Evaluating Digital Pathology Platforms and AI Algorithms for Scoring Consistency
Technical Support Center
FAQs & Troubleshooting Guides
Q1: Our AI model shows high scoring consistency on internal data but fails to generalize to slides from a different site. What are the primary causes? A: This is a common challenge in multi-site reproducibility research. The primary causes are typically:
- Pre-analytical Variability: Differences in tissue fixation, processing, and sectioning between sites.
- Scanner Variability: Disparities in slide scanner models, calibration, and imaging conditions (e.g., magnification, exposure).
- Staining Batch Effects: Inter- and intra-batch variability in IHC staining intensity and protocol execution.
- Algorithm Overfitting: The AI was trained on features specific to your site's "signature" (e.g., color profile, background hue) rather than biologically relevant features.
Troubleshooting Guide:
- Implement a Stain Normalization Pre-processing Step: Use algorithms (e.g., Reinhard, Macenko, Structure-Preserving Color Normalization) on all whole slide images (WSIs) before analysis.
- Create a Reference Slide Set: Circulate a set of physical tissue slides for all participating sites to stain and scan. Use this to quantify and correct for inter-site technical variance.
- Retrain with Multi-Site Data: Fine-tune your model using a small, carefully curated dataset from each new site to improve robustness.
Q2: When comparing two digital pathology platforms for IHC scoring, how should we design an experiment to evaluate platform-induced variability? A: Design a controlled, cross-platform comparison experiment.
Experimental Protocol:
- Sample Set: Select 30-50 representative IHC-stained tissue sections covering the full range of expected biomarker expression (negative, weak, moderate, strong).
- Scanning: Scan each physical slide on both Platform A and Platform B, using each platform's recommended default settings for clinical/research use. Ensure scanners are recently calibrated.
- Region of Interest (ROI) Annotation: A senior pathologist should annotate identical, matched ROIs on the paired digital images from both platforms.
- Analysis: Run the same AI scoring algorithm on the matched ROI pairs. Also, collect manual H-scores from 3 readers for each ROI on both platforms.
- Statistical Comparison: Calculate Intra-class Correlation Coefficient (ICC) and Bland-Altman limits of agreement for:
- Algorithm scores on Platform A vs. Platform B.
- Manual scores on Platform A vs. Platform B.
- Algorithm vs. Manual consensus for each platform separately.
Quantitative Data Summary: Table: Example Results Framework for Platform Comparison (Hypothetical Data)
| Comparison Metric | ICC (95% CI) | Bland-Altman Mean Difference (95% LoA) | Conclusion |
|---|---|---|---|
| AI Score: Platform A vs B | 0.87 (0.76, 0.93) | +0.15 H-score units (-12.5, +12.8) | Good agreement, minimal bias. |
| Manual Score: Platform A vs B | 0.92 (0.85, 0.96) | -2.1 H-score units (-18.3, +14.1) | Excellent agreement. |
| AI vs Manual on Platform A | 0.89 (0.80, 0.94) | +5.3 H-score units (-15.7, +26.3) | Good correlation, AI shows positive bias. |
| AI vs Manual on Platform B | 0.85 (0.73, 0.92) | +7.1 H-score units (-20.1, +34.3) | Good correlation, bias slightly higher. |
Q3: How do we validate that an AI algorithm's scoring consistency is acceptable for use in a multi-site drug development study? A: Follow a phased validation protocol aligned with regulatory guidelines (e.g., FDA AI/ML Software as a Medical Device).
Experimental Protocol: Analytical Validation
- Repeatability: The same user rescans 20 slides 3 times on the same scanner within a day. AI scores must have ICC > 0.95.
- Reproducibility:
- Inter-site: Scan 50 slides across 3 different sites (different scanner models). Calculate ICC between site averages. Target ICC > 0.90.
- Inter-algorithm: Compare scores from your AI against a validated, different method (e.g., manual scoring by central read committee). Establish equivalence limits.
- Linearity & Range: Use a cell line microarray or tissue cohort with known, graded expression levels. AI scores should show a linear correlation (R² > 0.85) with expected values.
- Robustness: Introduce controlled variations (e.g., 10% focus offset, mild stain color shift via software). The AI score change should not exceed pre-defined limits (e.g., ±5% H-score).
Visualization: Multi-Site AI Validation Workflow
Title: AI Validation Workflow for Multi-Site Studies
Visualization: Key Technical Variability Factors in IHC AI
Title: Factors Impacting IHC AI Scoring Consistency
The Scientist's Toolkit: Research Reagent & Material Solutions
Table: Essential Materials for Multi-Site IHC AI Validation Studies
| Item | Function & Rationale |
|---|---|
| Multi-Tissue Microarray (TMA) | Contains multiple tissue cores with varying expression levels on one slide. Enables highly controlled, high-throughput analysis of staining and AI performance across many tissues simultaneously. |
| Validated Reference Antibody | A primary antibody with established specificity and optimized concentration for the target biomarker. Critical for minimizing inter-site staining variability. |
| Automated IHC Stainer | Use of identical make/model or standardized protocols across sites reduces staining procedure variability, a major pre-analytical confounder. |
| Whole Slide Image (WSI) Scanner | High-resolution digital scanner. Calibration and maintenance logs must be kept. Using the same model at all sites is ideal. |
| Stain Normalization Software | Computational tool (e.g., OpenCV/CUDA-enabled libraries) to standardize color appearance of WSIs from different sites/scanners before AI analysis. |
| Digital Pathology Platform | Software ecosystem for viewing, managing, annotating, and analyzing WSIs. Must support integration of custom AI models and data export for statistical analysis. |
| Pathologist-Annotated Gold Standard Set | A set of WSIs with biomarker scores (e.g., H-score, % positivity) agreed upon by a panel of expert pathologists. Serves as the ground truth for AI training and validation. |
| Statistical Software (R, Python) | For calculating agreement metrics (ICC, Cohen's Kappa), generating Bland-Altman plots, and performing regression analysis to assess algorithm performance. |
Technical Support Center
FAQs & Troubleshooting
Q1: Our IHC assay passes internal validation but consistently receives low scores in NORDQC. What are the first parameters to investigate? A: Focus on pre-analytical variables. NORDQC data shows >70% of inter-laboratory variance stems from pre-analytical steps.
- Fixation: Adhere strictly to the recommended 6-24 hours in 10% NBF. Over-fixation (>48 hours) is a leading cause of false-negative results for many antigens.
- Antigen Retrieval: Verify pH and buffer composition. Re-test with both high-pH (e.g., Tris-EDTA, pH 9.0) and low-pH (e.g., citrate, pH 6.0) retrieval solutions. Document the exact heating time and cooling time.
- Control Tissue: Ensure your in-house positive control tissue matches the NORDQC sample type (e.g., tonsil for CD20).
Q2: How do we interpret discrepant results between CAP and NORDQC for the same biomarker? A: Analyze the proficiency testing (PT) design. CAP PT often uses curated tissue microarrays, while NORDQC typically employs whole-slide sections mimicking real-world diagnostics. Discrepancies often highlight pre-analytical heterogeneity.
- Actionable Protocol: Run a concordance experiment. Stain your CAP TMA slide and a full section from the same donor block with the same protocol. Compare staining intensity and homogeneity using quantitative image analysis (QIA) software. Tabulate the H-score variance.
Q3: Our automated staining platform shows high intra-site reproducibility but fails external PT. What is the likely culprit? A: This points to platform-specific protocol translation errors. The most common issue is the incorrect calculation of reagent volumes leading to inadequate coverage or drying.
- Troubleshooting Guide:
- Calculate Reaction Volume: Volume (µL) = Slide area (mm²) * Recommended cover thickness (mm). For a standard 15mm x 15mm cover, use ~150µL.
- Verify Dispense Height: Ensure the dispenser tip is <1mm above the liquid surface to prevent splashing.
- Check for Edge Dry-Out: Use a hydrophobic barrier pen or confirm the instrument's liquid containment settings.
Q4: What is the definitive method to confirm suspected antigen degradation in a PT sample? A: Implement a multiplexed internal control assay.
- Experimental Protocol: Stain the PT sample with a cocktail of antibodies targeting:
- The antigen of interest (e.g., ER, Clone SP1).
- A ubiquitous structural protein (e.g., Beta-actin, Clone AC-15).
- A nuclear marker (e.g., Histone H3).
- Analysis: If beta-actin and H3 staining are robust but the target antigen is negative, it confirms specific antigen loss. If all markers are weak, it indicates generalized sample degradation.
Data Presentation: Proficiency Testing Program Comparison
| Feature | NORDQC | CAP (College of American Pathologists) | UK NEQAS | GCP (Good Clinical Practice) Audit Focus |
|---|---|---|---|---|
| Primary Focus | Inter-laboratory reproducibility in diagnostics | Clinical laboratory accreditation | Comprehensive IHC & ISH quality | Assay robustness for multi-site clinical trials |
| Sample Type | Whole tissue sections | Tissue microarrays (TMAs) & whole sections | TMAs, cell lines, whole sections | Anonymized patient samples from trial sites |
| Key Metric | Staining intensity, specificity, heterogeneity | Pass/Fail against reference lab consensus | Quantitative score (0-8 scale) | Protocol deviation rate & concordance rate |
| Frequency | Bimonthly runs | Multiple surveys per year | Monthly to quarterly | Per clinical trial protocol (pre-study & ongoing) |
| Quantitative Data (2023) | Avg. participant concordance: 85-92% | >99% of labs pass major biomarkers | >90% of labs achieve satisfactory score (≥5) | Target intra-site CV <20%, inter-site CV <30% |
Experimental Protocols
Protocol 1: Establishing a Site-Specific Proficiency Testing Module Objective: To create an internal PT program mimicking NORDQC for pre-study assay training. Methodology:
- Select a well-characterized, multi-tissue block (e.g., tonsil, carcinoma, normal organ).
- Generate 40 serial sections. Randomly assign 5 sections to each of 5 scientists across 2 sites.
- Each scientist performs the IHC assay for a target (e.g., PD-L1, Clone 22C3) following the same SOP.
- A central pathologist scores all slides blinded using the NORDQC 0-3+ scale.
- Calculate the inter-operator and inter-site Cohen's kappa coefficient. Target κ > 0.8.
Protocol 2: Titration of Primary Antibody Using PT Reference Material Objective: To determine the optimal antibody concentration that matches the consensus staining pattern of a PT sample. Methodology:
- Obtain a residual section from a past NORDQC sample (e.g., HER2 challenge).
- Prepare a dilution series of the primary antibody (e.g., 1:50, 1:100, 1:200, 1:500, 1:1000).
- Perform IHC under otherwise identical conditions.
- Score staining intensity and membranous completeness. Select the dilution that yields a 3+ score in the known positive cell population without background in negative areas. This becomes your site's standardized concentration for that antibody lot.
Mandatory Visualizations
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in IHC PT Benchmarking |
|---|---|
| Multitissue Microarray (MTA) Block | Contains control tissues for multiple antigens; used for daily run validation and antibody titration. |
| ER/PR/Her2 Control Cell Lines | Commercially available pellets with known score (0, 1+, 2+, 3+); essential for quantitative assay calibration. |
| Chromogenic Detection Kit (Polymer-based) | Amplifies signal with high sensitivity and low background; key for standardizing detection across sites. |
| Antigen Retrieval Buffer (pH 6.0 & 9.0) | Unmasks epitopes; having both pH options is critical for troubleshooting PT failures. |
| Hydrophobic Barrier Pen | Creates a barrier around tissue sections to prevent reagent spread and edge drying during manual staining. |
| Digital Image Analysis Software | Enables quantitative scoring (H-score, % positivity); reduces inter-observer variability for PT review. |
| Reference Antibody Panel | WHO/IFCC recommended antibody clones (e.g., ER/SP1, HER2/4B5) for cross-referencing in-house reagents. |
Conclusion
Achieving robust, reproducible IHC across multiple sites is not merely a technical hurdle but a fundamental requirement for credible translational research and successful drug development. By addressing foundational variables, implementing rigorous methodological frameworks, proactively troubleshooting discrepancies, and employing comprehensive validation, teams can transform IHC from a subjective art into a reliable, quantitative science. The future lies in further integration of digital pathology, artificial intelligence for standardized scoring, and global adoption of unified SOPs and reference materials. Embracing these principles will enhance data integrity, accelerate biomarker discovery, and ultimately increase the success rate of clinical trials, bridging the gap between bench findings and patient benefit.