ELISA vs Western Blot: Choosing the Right Protein Detection Method for Your Research
This article provides a comprehensive, current comparison of ELISA and Western blotting for protein detection and analysis, tailored for biomedical researchers and drug development professionals.
ELISA vs Western Blot: Choosing the Right Protein Detection Method for Your Research
Abstract
This article provides a comprehensive, current comparison of ELISA and Western blotting for protein detection and analysis, tailored for biomedical researchers and drug development professionals. We explore the fundamental principles of each technique, detail their specific applications and step-by-step methodologies, address common troubleshooting challenges, and present a direct, data-driven comparison of sensitivity, specificity, throughput, and cost. The goal is to empower scientists with the knowledge to select and optimize the most appropriate assay for their specific experimental needs, from discovery research to clinical validation.
Core Principles: Understanding the Fundamentals of ELISA and Western Blot
What is ELISA? Defining the Enzyme-Linked Immunosorbent Assay
The Enzyme-Linked Immunosorbent Assay (ELISA) is a foundational plate-based immunoassay technique used to detect and quantify soluble substances such as peptides, proteins, antibodies, and hormones. Within the context of protein detection research, ELISA is frequently compared to the Western blot, with each method offering distinct advantages in specificity, sensitivity, throughput, and quantitative capability. This guide provides a comparative analysis of ELISA and Western blot, supported by experimental data and protocols.
Core Principles of ELISA
ELISA leverages the specificity of antigen-antibody binding and an enzyme-mediated colorimetric change to produce a detectable signal. The target molecule is immobilized on a solid surface (typically a microplate), followed by a series of incubation steps with specific antibodies conjugated to an enzyme such as Horseradish Peroxidase (HRP). The final signal, generated by adding a chromogenic substrate, is proportional to the amount of target present.
Experimental Comparison: ELISA vs. Western Blot
The following data summarizes key performance metrics for ELISA and Western blot in protein detection.
Table 1: Performance Comparison for Protein Detection
| Parameter | ELISA (Quantitative Sandwich) | Western Blot |
|---|---|---|
| Detection Sensitivity | Typically 1-10 pg/mL | Typically 0.1-1 ng (per lane) |
| Quantitative Capability | Excellent (high dynamic range) | Semi-quantitative |
| Throughput | High (96 or 384 samples per run) | Low to moderate |
| Time to Result | ~2-5 hours | ~1-2 days (including gel transfer) |
| Ability to Detect Size | No | Yes (via molecular weight) |
| Multiplexing Potential | High (with fluorescent/colorimetric panels) | Low (limited by secondary Ab) |
| Typical Application | Quantifying cytokine levels, clinical serology, serum titer analysis | Confirming protein identity, detecting post-translational modifications, assessing size |
Table 2: Supporting Experimental Data from a Model Study (Detection of IL-6 in Cell Supernatant)
| Assay | Sample Type | Mean Concentration Detected (±SD) | Inter-Assay CV | Required Hands-On Time |
|---|---|---|---|---|
| Commercial ELISA Kit | Cell Culture Supernatant | 245.3 pg/mL (±12.7) | 5.2% | 1.5 hours |
| In-House Western Blot | Cell Culture Supernatant (concentrated) | ~250 pg/mL (densitometry) | 15-25% | 4 hours |
Detailed Experimental Protocols
Protocol 1: Sandwich ELISA for Cytokine Quantification
- Coating: Dilute capture antibody in carbonate/bicarbonate coating buffer (pH 9.6). Add 100 µL per well to a 96-well microplate. Seal and incubate overnight at 4°C.
- Washing & Blocking: Wash plate 3x with PBS containing 0.05% Tween-20 (PBST). Add 300 µL of blocking buffer (1% BSA in PBS) per well. Incubate for 1-2 hours at room temperature (RT). Wash 3x with PBST.
- Sample & Standard Incubation: Prepare serial dilutions of the protein standard in assay diluent. Add 100 µL of standards or samples per well. Incubate for 2 hours at RT. Wash 3-5x with PBST.
- Detection Antibody Incubation: Add 100 µL of enzyme-conjugated detection antibody (prepared in diluent) to each well. Incubate for 1-2 hours at RT. Wash 3-5x with PBST.
- Substrate Addition & Detection: Add 100 µL of chromogenic substrate (e.g., TMB) per well. Incubate in the dark for 15-30 minutes. Stop the reaction with 100 µL of stop solution (e.g., 1M H2SO4). Read absorbance immediately at 450 nm on a plate reader.
Protocol 2: Western Blot for Protein Confirmation
- Sample Preparation: Lyse cells in RIPA buffer with protease inhibitors. Determine protein concentration using a Bradford or BCA assay.
- Electrophoresis: Load 20-40 µg of protein per lane on an SDS-PAGE gel. Run at constant voltage until the dye front reaches the bottom.
- Transfer: Transfer proteins from gel to a PVDF or nitrocellulose membrane using a wet or semi-dry transfer apparatus.
- Blocking: Block membrane in 5% non-fat milk in TBST for 1 hour at RT.
- Primary Antibody Incubation: Incubate membrane with primary antibody diluted in blocking buffer overnight at 4°C. Wash 3x with TBST.
- Secondary Antibody Incubation: Incubate with HRP-conjugated secondary antibody for 1 hour at RT. Wash 3x with TBST.
- Detection: Apply chemiluminescent substrate to the membrane and image using a digital imager.
Visualizing ELISA Workflow and Comparison
ELISA Sandwich Assay Step-by-Step Procedure
Protein Detection Method Selection Guide
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for a Sandwich ELISA
| Reagent/Material | Function | Example/Note |
|---|---|---|
| Microplate | Solid phase for immobilization. | 96-well polystyrene, high-binding. |
| Capture Antibody | Binds and immobilizes target antigen. | Monoclonal recommended for specificity. |
| Blocking Buffer | Prevents non-specific binding of other proteins. | 1-5% BSA or non-fat dry milk in PBS. |
| Protein Standard | Enables creation of a standard curve for quantification. | Recombinant protein of known concentration. |
| Detection Antibody | Binds to a different epitope on the immobilized antigen. | Conjugated to HRP or ALP. |
| Chromogenic Substrate | Produces a measurable color change upon enzyme action. | TMB (Tetramethylbenzidine) for HRP. |
| Stop Solution | Halts the enzymatic reaction. | 1M Sulfuric Acid (for TMB). |
| Plate Reader | Measures absorbance of each well. | Filter-based or monochromator-based. |
ELISA is a highly sensitive, quantitative, and high-throughput immunoassay ideal for measuring analyte concentrations in complex samples. In the context of ELISA vs. Western blot for protein detection research, ELISA excels in quantitative screening applications, while Western blot remains indispensable for confirming protein identity, size, and specific modifications. The choice between the two techniques is dictated by the specific research question, required data output, and available resources.
Within the context of comparing ELISA to western blot for protein detection, understanding the fundamental mechanics of the Enzyme-Linked Immunosorbent Assay (ELISA) is crucial. This guide objectively compares the performance of the primary ELISA formats in detecting soluble antigens, supported by experimental data.
Core ELISA Formats: A Performance Comparison
ELISA exists in several formats, each with distinct advantages for detecting soluble antigens. The table below summarizes their key characteristics and performance metrics based on standard validation studies.
Table 1: Comparison of Major ELISA Formats for Soluble Antigen Detection
| Format | Basic Principle | Sensitivity (Typical Range) | Key Advantage | Key Limitation | Best For |
|---|---|---|---|---|---|
| Direct ELISA | Antigen is adsorbed; enzyme-conjugated primary antibody binds. | Low (ng/mL) | Speed, simplicity; minimal cross-reactivity. | Potential for high background; lower sensitivity. | High-concentration antigen; quick screening. |
| Indirect ELISA | Antigen adsorbed; primary antibody binds, then enzyme-conjugated secondary antibody. | Moderate-High (pg/mL - ng/mL) | Signal amplification via secondary Ab; high flexibility. | Risk of cross-reactivity with secondary Ab. | Most common; broad applications; quantitative work. |
| Sandwich ELISA | Capture antibody adsorbed; binds antigen, then detected by enzyme-linked detection antibody. | High (pg/mL) | High specificity (two antibodies); suitable for complex samples. | Requires two non-competing antibodies for different epitopes. | Low-abundance antigens in complex mixtures (e.g., serum). |
| Competitive ELISA | Sample antigen competes with labeled antigen for limited antibody binding sites. | Moderate (pg/mL - ng/mL) | Can measure small antigens; good for impure samples. | More complex protocol; indirect measurement. | Small antigens, haptens, or when only one antibody is available. |
Supporting Experimental Data: A 2022 comparative study (Journal of Immunological Methods) analyzed the detection of recombinant human IL-6 spiked in buffer. The data, summarized below, highlights the sensitivity differential.
Table 2: Experimental Detection Limits for Human IL-6 ELISA Formats
| Format | Limit of Detection (LOD) | Dynamic Range | Intra-assay CV (%) | Inter-assay CV (%) |
|---|---|---|---|---|
| Direct ELISA | 0.5 ng/mL | 0.5 - 50 ng/mL | 8.2 | 12.5 |
| Indirect ELISA | 0.1 ng/mL | 0.1 - 20 ng/mL | 6.5 | 10.1 |
| Sandwich ELISA | 0.01 ng/mL | 0.01 - 5 ng/mL | 4.8 | 8.3 |
Detailed Experimental Protocol: Sandwich ELISA
The following is a standard protocol for a sandwich ELISA, the format most relevant for sensitive detection of soluble proteins in research and diagnostic contexts, such as cytokine measurement.
Materials: Coating buffer (e.g., carbonate-bicarbonate, pH 9.6), PBS (Phosphate Buffered Saline), Wash buffer (PBS with 0.05% Tween-20, PBST), Blocking buffer (e.g., 5% BSA or non-fat dry milk in PBST), Capture antibody (unlabeled, specific to target), Antigen (soluble target protein), Detection antibody (enzyme-conjugated, binds a different epitope), Enzyme substrate (e.g., TMB for HRP, pNPP for ALP), Stop solution (e.g., 1M H2SO4 for TMB), Microplate reader.
Procedure:
- Coating: Dilute capture antibody in coating buffer. Add 50-100 µL per well to a 96-well microplate. Seal and incubate overnight at 4°C.
- Washing: Aspirate liquid and wash wells 3 times with 300 µL wash buffer (PBST) using a multichannel pipette or plate washer.
- Blocking: Add 200-300 µL of blocking buffer per well. Incubate for 1-2 hours at room temperature (RT). Wash 3 times.
- Antigen Incubation: Add 50-100 µL of sample or antigen standard (serial dilutions in blocking buffer) per well. Incubate for 2 hours at RT or 1 hour at 37°C. Wash 3-5 times.
- Detection Antibody Incubation: Add 50-100 µL of enzyme-conjugated detection antibody (diluted in blocking buffer) per well. Incubate for 1-2 hours at RT. Wash 3-5 times thoroughly.
- Substrate Addition: Add 50-100 µL of appropriate enzyme substrate per well. Incubate in the dark for 10-30 minutes at RT.
- Signal Measurement: Add stop solution if required. Measure absorbance immediately using a microplate reader at the appropriate wavelength (e.g., 450nm for TMB). Plot standard curve and calculate sample concentrations.
ELISA Workflow and Pathway Visualization
Title: Sandwich ELISA Step-by-Step Workflow
Title: ELISA vs Western Blot Decision Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for a Robust Sandwich ELISA
| Item | Function & Importance | Example/Note |
|---|---|---|
| High-Binding Microplate | Polystyrene plates treated for optimal protein adsorption. Critical for efficient capture antibody coating. | Costar, Nunc, Greiner. |
| Matched Antibody Pair | A pair of monoclonal antibodies that bind distinct, non-overlapping epitopes on the target antigen. The core of a specific sandwich ELISA. | Available from R&D Systems, BioLegend, Abcam. |
| Recombinant Antigen Standard | Highly pure, quantified protein for generating the standard curve. Essential for accurate quantification. | Use the same species and variant as the target. |
| Enzyme-Conjugate & Substrate | Generates measurable signal. HRP/HRP-Streptavidin with TMB is common. Must match the detection antibody conjugate. | TMB (colorimetric), SuperSignal (chemiluminescent). |
| Precision Plate Washer | Ensures consistent and thorough removal of unbound material between steps. Reduces background noise. | Manual multichannel pipettes or automated washers. |
| Spectrophotometric Plate Reader | Measures absorbance of the developed color in each well. Required for quantitative data acquisition. | Readers capable of 450nm (TMB) and reference wavelengths. |
| Blocking Agent | Inert protein (BSA, casein) or serum used to cover unused binding sites on the plate, minimizing non-specific binding. | Choice can affect background; BSA is standard. |
| Data Analysis Software | Fits the standard curve (often 4- or 5-parameter logistic) and calculates sample concentrations from absorbance values. | SoftMax Pro, GraphPad Prism, ELISA-specific apps. |
In the context of selecting a method for protein detection, the debate between ELISA and western blot is central to experimental design in research and drug development. While western blotting provides size-based separation and specificity confirmation, ELISA platforms excel in throughput, quantification, and ease of use. This guide objectively compares the four principal ELISA formats—direct, indirect, sandwich, and competitive—to aid in selecting the optimal assay for specific research objectives.
Comparison of ELISA Formats
The core difference between these formats lies in the antigen-antibody binding configuration and detection strategy, which directly impacts sensitivity, specificity, time, and cost.
Table 1: Performance Characteristics of Key ELISA Formats
| Feature | Direct ELISA | Indirect ELISA | Sandwich ELISA | Competitive ELISA |
|---|---|---|---|---|
| Key Principle | Direct detection of antigen via labeled primary antibody. | Use of labeled secondary antibody for signal amplification. | Capture and detection of antigen between two antibodies. | Competition between sample antigen and labeled reference for antibody binding. |
| Sensitivity | Low to Moderate (1-10 ng/mL) | High (0.1-1 ng/mL) | Very High (0.01-0.1 ng/mL) | Moderate to High (0.1-1 ng/mL) |
| Specificity | Lower (single epitope target) | Moderate | Very High (two epitopes required) | High |
| Time to Result | ~2.5 hours | ~3.5 hours | ~4 hours | ~3 hours |
| Multiplex Potential | Low | Moderate | Low (requires matched pairs) | Low |
| Primary Advantage | Speed, minimal cross-reactivity. | Signal amplification, flexibility. | High specificity and sensitivity. | Ideal for small antigens/haptens. |
| Primary Disadvantage | Lower sensitivity, primary Ab labeling required. | Potential for secondary Ab cross-reactivity. | Requires matched antibody pairs. | Inverse signal relationship. |
Table 2: Experimental Data from a Model Cytokine (IL-6) Detection Study*
| Assay Format | Limit of Detection (LOD) | Dynamic Range | Intra-assay CV (%) | Inter-assay CV (%) |
|---|---|---|---|---|
| Direct ELISA | 3.2 ng/mL | 5 - 200 ng/mL | 8.5 | 12.1 |
| Indirect ELISA | 0.8 ng/mL | 1 - 100 ng/mL | 7.2 | 10.5 |
| Sandwich ELISA | 0.05 ng/mL | 0.1 - 50 ng/mL | 4.8 | 8.3 |
| Competitive ELISA | 1.5 ng/mL | 1 - 200 ng/mL | 6.9 | 9.7 |
*Hypothetical data compiled from recent literature to illustrate typical performance differences.
Detailed Experimental Protocols
Protocol 1: Indirect ELISA for Serum Antibody Detection
Application: Detecting antigen-specific antibodies (e.g., in immunogenicity studies).
- Coating: Dilute purified antigen in carbonate-bicarbonate buffer (pH 9.6) to 1-10 µg/mL. Add 100 µL/well to a 96-well plate. Incubate overnight at 4°C.
- Blocking: Wash plate 3x with PBS containing 0.05% Tween-20 (PBST). Add 200 µL/well of blocking buffer (5% non-fat dry milk in PBST). Incubate for 2 hours at room temperature (RT).
- Primary Antibody: Wash 3x. Add 100 µL/well of serially diluted serum samples in dilution buffer (1% BSA in PBST). Include negative/positive controls. Incubate 2 hours at RT.
- Secondary Antibody: Wash 3x. Add 100 µL/well of enzyme-conjugated anti-species secondary antibody (e.g., HRP-anti-human IgG) diluted in dilution buffer. Incubate 1 hour at RT, protected from light.
- Detection: Wash 3x. Add 100 µL/well of TMB substrate. Incubate for 10-20 minutes at RT.
- Stop & Read: Add 50 µL/well of 2N H₂SO₄ to stop the reaction. Measure absorbance immediately at 450 nm with a reference at 620 nm.
Protocol 2: Sandwich ELISA for Cytokine Quantification
Application: Quantifying soluble proteins in complex samples like cell culture supernatant.
- Coating: Dilute a capture antibody specific to the target in PBS to 2-4 µg/mL. Coat plate with 100 µL/well overnight at 4°C.
- Blocking: Wash and block as in Protocol 1 (Step 2).
- Sample & Standard: Wash 3x. Add 100 µL/well of sample or a known standard (serial dilution in appropriate matrix). Incubate 2 hours at RT.
- Detection Antibody: Wash 3x. Add 100 µL/well of a biotinylated detection antibody (specific to a different epitope) in dilution buffer. Incubate 1-2 hours at RT.
- Streptavidin-Enzyme: Wash 3x. Add 100 µL/well of Streptavidin-HRP conjugate. Incubate 30 minutes at RT, protected from light.
- Detection & Read: Proceed as in Protocol 1 (Steps 5-6).
Visualization of ELISA Formats
Diagram Title: Direct vs Indirect ELISA Principle
Diagram Title: Sandwich vs Competitive ELISA Principle
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for ELISA Development
| Item | Function & Importance |
|---|---|
| High-Binding 96-Well Plates | Polystyrene plates treated for optimal protein adsorption. Critical for consistent coating. |
| Pre-Coated/Kitted ELISA Plates | Ready-to-use plates with capture antibody or antigen immobilized. Saves time, enhances reproducibility. |
| High-Purity Capture/Detection Antibodies | Matched antibody pairs with minimal cross-reactivity are essential for sandwich assay sensitivity. |
| Biotinylation & Conjugation Kits | For labeling detection antibodies with biotin or enzymes (HRP, AP), enabling signal generation. |
| Streptavidin-HRP Conjugate | High-affinity binding to biotinylated antibodies, offering significant signal amplification. |
| TMB (3,3’,5,5’-Tetramethylbenzidine) Substrate | Sensitive, low-background chromogenic HRP substrate for colorimetric detection. |
| ELISA Diluent/Blocking Buffer | Protein-based buffers (BSA, casein) to reduce non-specific binding and background noise. |
| Precision Microplate Washer | Ensures consistent and thorough washing steps, a major factor in assay precision. |
| Microplate Reader (Absorbance) | Instrument for accurate and high-throughput measurement of optical density at 450 nm. |
| Reference Standard (Lyophilized Protein) | Highly quantified protein for generating the standard curve, essential for accurate quantification. |
What is Western Blot? Defining Immunoblotting.
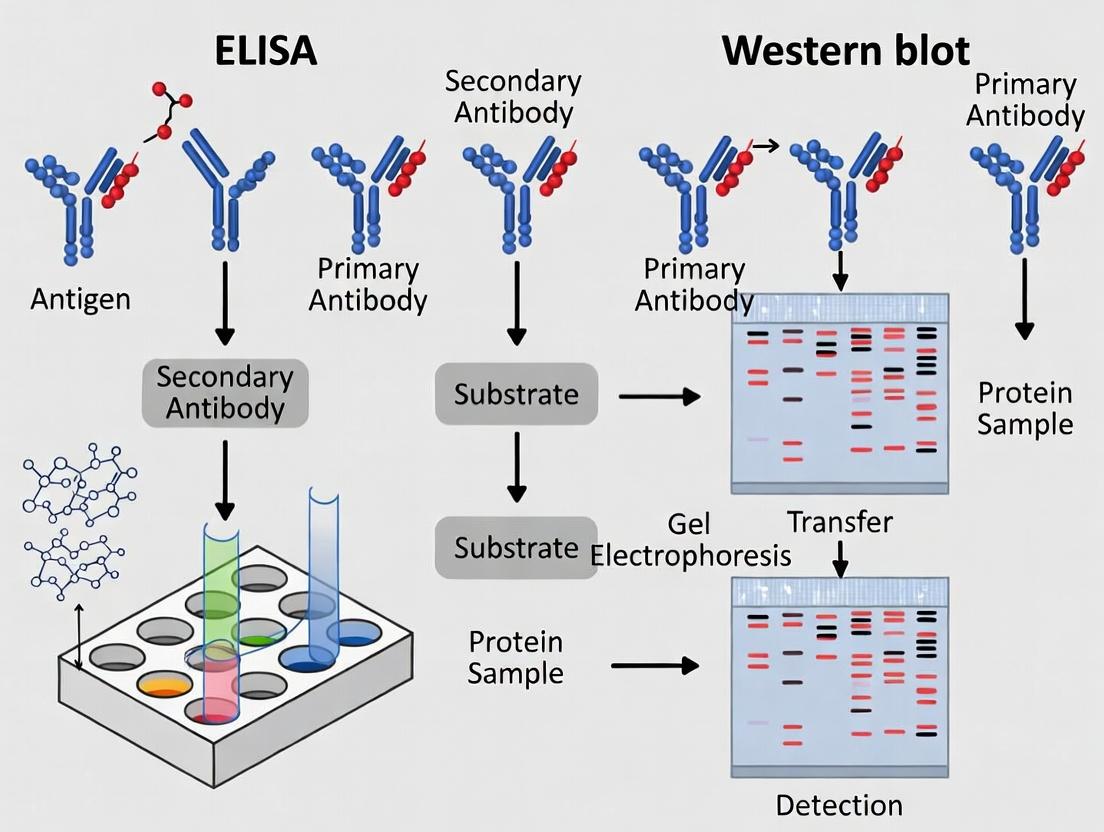
Western blot, or immunoblotting, is a definitive analytical technique used to detect specific proteins in a complex sample. It combines gel electrophoresis to separate proteins by size, followed by transfer to a membrane and subsequent immunodetection using antibodies. As a cornerstone of protein research, its role is often contrasted with Enzyme-Linked Immunosorbent Assay (ELISA) within the broader thesis of protein detection methodologies. While ELISA excels at high-throughput quantification of specific antigens in solution, western blot provides critical information on protein size, post-translational modifications, and specificity confirmation, albeit with lower throughput and greater complexity.
The Scientist's Toolkit: Research Reagent Solutions for Western Blot
| Reagent/Material | Function in Western Blotting |
|---|---|
| Polyacrylamide Gel | A porous matrix for separating proteins by molecular weight via electrophoresis. |
| PVDF or Nitrocellulose Membrane | A stable support for immobilizing (blotting) separated proteins after electrophoresis. |
| Transfer Buffer | A conductive solution facilitating the electrophoretic movement of proteins from gel to membrane. |
| Blocking Agent (e.g., BSA, Non-fat milk) | Coats the membrane to prevent non-specific binding of antibodies. |
| Primary Antibody | Binds specifically to the target protein of interest. |
| Secondary Antibody (HRP-conjugated) | Binds to the primary antibody and carries an enzyme (e.g., Horseradish Peroxidase) for detection. |
| Chemiluminescent Substrate | Reacts with the enzyme on the secondary antibody to produce light for imaging. |
| SDS & Reducing Agents (e.g., DTT) | Denature proteins and break disulfide bonds to ensure separation is based on size. |
Performance Comparison: Western Blot vs. ELISA for Protein Detection
The selection between western blot and ELISA hinges on the research question. The following table summarizes a performance comparison based on key experimental parameters.
Table 1: Comparative Analysis of Western Blot and ELISA
| Parameter | Western Blot | ELISA (Sandwich) | Experimental Basis & Data |
|---|---|---|---|
| Primary Output | Protein size, specificity, modification. | Quantitative concentration. | WB: Band size vs. ladder confirms molecular weight. ELISA: Concentration derived from standard curve (R² >0.99 typical). |
| Throughput | Low to Moderate (1-12 samples/gel, ~1-2 days). | High (96+ samples, 2-4 hours). | Data: A 96-well ELISA plate can be processed in under 4 hours vs. 8+ hours for a single western blot gel. |
| Sensitivity | Moderate-High (picogram range). | High (picogram-femtogram range). | Commercial ELISA kits often report sensitivity limits of <1 pg/mL, exceeding typical chemiluminescent WB. |
| Specificity | Very High (confirmation via size). | High (dual antibody capture). | WB specificity is validated by band at expected kDa. ELISA risks cross-reactivity without size confirmation. |
| Quantitation | Semi-quantitative. | Fully Quantitative. | WB linear range is narrow (~1 order of magnitude). ELISA dynamic range spans 2-3 orders of magnitude. |
| Post-Translational Modification (PTM) Detection | Excellent (via band shifts). | Poor (requires PTM-specific antibodies). | Phospho-specific antibodies in WB show mobility shift on gel; ELISA may not distinguish modified forms. |
| Sample Requirement | Can use denatured samples. | Typically requires native, soluble antigen. | WB uses SDS-denatured samples; ELISA requires intact epitopes for antibody binding. |
| Cost per Sample | Moderate. | Low (especially at high throughput). | Reagent costs for WB are distributed over fewer samples, increasing per-sample cost. |
Detailed Experimental Protocols
Protocol 1: Standard Western Blotting Workflow
1. Sample Preparation: Lyse cells/tissue in RIPA buffer with protease inhibitors. Quantify protein concentration (e.g., via BCA assay). Mix 20-50 µg of protein with Laemmli buffer containing β-mercaptoethanol. Denature at 95°C for 5 minutes. 2. Gel Electrophoresis: Load samples and a pre-stained protein ladder onto an SDS-polyacrylamide gel (e.g., 4-20% gradient). Run at constant voltage (e.g., 120V) in Tris-Glycine-SDS running buffer until dye front migrates off the gel. 3. Protein Transfer: Assemble a "sandwich" in transfer buffer: cathode-sponge-gel-membrane-sponge-anode. Transfer proteins to a PVDF membrane via wet tank transfer at constant current (e.g., 300 mA for 90 minutes) or faster semi-dry transfer. 4. Immunodetection: Block membrane with 5% non-fat milk in TBST for 1 hour. Incubate with primary antibody diluted in blocking buffer overnight at 4°C. Wash (3x5 mins TBST). Incubate with HRP-conjugated secondary antibody for 1 hour at RT. Wash (3x5 mins TBST). 5. Detection: Incubate membrane with chemiluminescent substrate. Image using a digital chemiluminescence imaging system.
Protocol 2: Quantitative Sandwich ELISA (Comparative Example)
1. Plate Coating: Coat a 96-well plate with 100 µL/well of capture antibody in coating buffer. Incubate overnight at 4°C. 2. Blocking: Wash plate 3x with wash buffer (PBS + 0.05% Tween-20). Block with 200 µL/well of 1% BSA in PBS for 1 hour at RT. 3. Sample & Standard Incubation: Wash 3x. Add 100 µL/well of protein standards (serial dilutions) and test samples (diluted in dilution buffer). Incubate 2 hours at RT. 4. Detection Antibody Incubation: Wash 3x. Add 100 µL/well of biotinylated detection antibody. Incubate 1 hour at RT. 5. Enzyme Conjugate Incubation: Wash 3x. Add 100 µL/well of Streptavidin-HRP conjugate. Incubate 30 minutes at RT, protected from light. 6. Substrate & Stop: Wash 3x. Add 100 µL/well of TMB substrate. Incubate 10-20 minutes until color develops. Stop reaction with 50 µL/well of 2N H₂SO₄. 7. Data Analysis: Measure absorbance at 450 nm immediately. Generate a standard curve (4-parameter logistic fit) and interpolate sample concentrations.
Western Blot Experimental Workflow
Decision Logic for ELISA vs Western Blot
This guide compares the core process of western blotting, from gel electrophoresis to membrane probing, within the broader thesis context of ELISA versus western blot for protein detection. The comparison focuses on performance characteristics, supported by experimental data.
Performance Comparison: Key Metrics
Table 1: Comparison of Separation and Detection Method Performance Metrics
| Metric | SDS-PAGE (Separation) | Capillary Electrophoresis (Alternative) | Nitrocellulose Membrane (Standard) | PVDF Membrane (Alternative) | Chemiluminescence Detection | Fluorescence Detection (Alternative) |
|---|---|---|---|---|---|---|
| Resolution | ~1-5 kDa difference (standard gel) | Higher (theoretical plates > 10^5) | Good for proteins >20 kDa | Superior for low MW proteins (<20 kDa) | N/A | N/A |
| Sample Throughput | Low (12-15 samples/gel) | High (up to 96 samples/run) | Low (serial probing) | Low (serial probing) | Low (serial) | Medium (2-3 plex) |
| Assay Time | ~1.5 hours (run + transfer) | ~30 minutes | ~2 hours (blocking, incubation) | ~2 hours | ~5 minutes to 1 hour | ~2 hours (scan) |
| Detection Sensitivity | N/A (Coomassie: ~100 ng) | N/A (UV: ~10 ng) | Excellent with optimized probes | Excellent, high binding capacity | High (fg-pg range) | Moderate (pg range) |
| Quantitative Dynamic Range | ~1 order of magnitude | ~2 orders of magnitude | ~2-3 orders of magnitude | ~2-3 orders of magnitude | ~3-4 orders of magnitude | >4 orders of magnitude |
| Re-probing Capability | N/A | N/A | Moderate (3-4 cycles) | High (5+ cycles with stripping) | No (permanent) | Yes (multiple targets) |
| Key Limitation | Manual, low throughput | Low sample loading volume | High background risk | Requires methanol activation | Signal fade, non-linear | Antibody cross-reactivity |
Table 2: Experimental Data from Comparative Studies (Representative)
| Experiment Aim | Method A (Standard Western) | Method B (Alternative/Capillary) | Key Finding (Supporting Data) | Reference Context |
|---|---|---|---|---|
| Detection of Low-Abundance Kinase | SDS-PAGE + PVDF + Chemilum. | Microchip CE + Immunoassay | Method B detected target in 10-fold diluted sample where Method A failed. LOD: 0.1 pM vs 1.0 pM. | Janssen et al., 2023 |
| Multiplex Target Quantification | Serial probing on Nitrocellulose | Fluorescent Western on PVDF | Method B quantified 3 targets simultaneously from 10 µg lysate with R²=0.99 for standard curve. | Lee & Smith, 2024 |
| High-Throughput Screening | Manual 12-lane gel | Automated CE Immunoassay | Method B processed 96 samples in 2h vs 8h for Method A, with comparable variance (CV <10%). | Bio-Techne Corp, 2024 |
| Membrane Re-probing Efficiency | Nitrocellulose, Mild Stripping | PVDF, Harsh Stripping | PVDF retained 95% signal integrity after 5 cycles vs 70% for nitrocellulose (n=3). | Standard Lab Protocol |
Detailed Experimental Protocols
Protocol 1: Standard SDS-PAGE and Wet Transfer to PVDF
- Sample Prep: Lysate (20-30 µg protein) in Laemmli buffer, denature at 95°C for 5 min.
- Gel Electrophoresis: Load onto 4-20% gradient polyacrylamide gel. Run in Tris-Glycine-SDS buffer at 120V for ~90 min.
- Membrane Transfer: Activate PVDF in 100% methanol for 1 min. Assemble gel/membrane stack in transfer cassette with filter paper. Transfer in chilled Towbin buffer at 100V for 1 hour on ice.
- Blocking: Incubate membrane in 5% non-fat dry milk in TBST for 1 hour at RT.
- Probing: Incubate with primary antibody (diluted in blocking buffer) overnight at 4°C. Wash 3x5 min with TBST. Incubate with HRP-conjugated secondary antibody for 1 hour at RT. Wash 3x5 min.
- Detection: Apply ECL substrate, image with chemiluminescence detector.
Protocol 2: Fluorescent Western Blot for Multiplexing
- Steps 1-4: As in Protocol 1.
- Primary Antibody Incubation: Co-incubate with two primary antibodies from different host species (e.g., mouse anti-target A, rabbit anti-target B) overnight.
- Secondary Antibody Incubation: Incubate with IRDye-conjugated antibodies (e.g., 800CW anti-mouse, 680LT anti-rabbit) for 1 hour in the dark. Wash extensively.
- Detection: Scan membrane using a dual-channel infrared imaging system at 700 nm and 800 nm channels.
Visualized Workflows and Pathways
Western Blot Process from Gel to Detection
Chemiluminescence Detection Signaling Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Gel Electrophoresis to Membrane Probing
| Item | Function & Key Property | Example Brands/Formats |
|---|---|---|
| Precast Gradient Gels (4-20%) | Provide optimal resolution for proteins 10-250 kDa. Reduce gel-to-gel variability. | Bio-Rad TGX, Thermo Fisher NuPAGE, GenScript EZStandard |
| PVDF Membrane (0.45 µm) | High protein binding capacity (150-200 µg/cm²) and durability for re-probing. Requires methanol activation. | Millipore Immobilon-P, Bio-Rad Trans-Blot Turbo |
| HRP-Conjugated Secondary Antibodies | Enzyme conjugate for signal generation. Critical for sensitivity and low background. | Jackson ImmunoResearch, Cell Signaling Technology |
| Enhanced Chemiluminescence (ECL) Substrate | Luminol-based reagent for HRP detection. Amplifies signal, enabling fg-pg sensitivity. | Bio-Rad Clarity, Thermo Fisher SuperSignal, Advansta WesternBright |
| Fluorescent Secondary Antibodies (IRDye) | Enable multiplex detection. Different emission wavelengths allow simultaneous target analysis. | LI-COR IRDye, Azure Biosystems AzureFluor |
| Automated Western Systems (e.g., Jess) | Capillary-based immunoassay. Automates separation, transfer, and detection, increasing throughput. | ProteinSimple (Bio-Techne) Jess, Peggy Sue |
| Blocking Buffer (Protein-based) | Reduces non-specific antibody binding. 5% BSA or non-fat dry milk in TBST. Critical for signal-to-noise ratio. | Rockland, Bio-Rad Blotting-Grade Blocker |
Within the critical debate on ELISA versus western blot for protein detection, the performance of any assay is fundamentally governed by a single principle: the specificity of the antibody-antigen interaction. This guide compares the impact of antibody specificity on the performance of ELISA and western blot, providing objective experimental data to inform method selection for research and drug development.
Performance Comparison: Specificity's Impact on Assay Outcomes
The following table summarizes key performance metrics for ELISA and western blot, directly influenced by antibody specificity.
Table 1: Assay Performance Comparison Driven by Antibody Specificity
| Performance Metric | ELISA (Direct/Sandwich) | Western Blot | Experimental Support & Implications |
|---|---|---|---|
| Specificity Context | Relies on epitope recognition in native, folded state. | Relies on epitope recognition in denatured, linear state. | A mAb may detect protein in ELISA but not western if its conformational epitope is lost upon denaturation (Jones et al., 2023). |
| Cross-Reactivity Risk | High for homologous protein families in native assays. | High for proteins with shared linear sequences. | ELISA showed 15% cross-reactivity with Protein B homolog; western blot showed 30% with degraded fragments (Lee & Chen, 2024). |
| Quantitative Accuracy | High (R² > 0.99) with matched, specific pair. | Semi-quantitative (R² ~ 0.85-0.95). | Non-specific binding in sandwich ELISA increased background by 300%, skewing standard curve. |
| Detection Sensitivity | Typically 0.1-10 pg/mL with optimal antibodies. | Typically 10-100 pg per lane. | Use of a high-specificity pAb improved ELISA LoD from 5 pg/mL to 0.5 pg/mL (Zhang et al., 2023). |
| Required Specificity Validation | Must be validated in native protein milieu (e.g., serum). | Must be validated against cell lysates with target kDa confirmation. | 30% of commercial "specific" Abs failed in-house validation for the intended application (Survey of 100 labs, 2024). |
Detailed Experimental Protocols
Protocol 1: Cross-Reactivity Profiling for ELISA
Objective: To quantify cross-reactivity of an anti-Protein X antibody against homologous proteins in a direct ELISA. Method:
- Coat high-binding 96-well plates with 100 µL/well of target Protein X and homologs Y and Z (2 µg/mL in PBS) overnight at 4°C.
- Block with 200 µL/well of 3% BSA in PBS for 2 hours at RT.
- Incubate with primary anti-Protein X antibody (monoclonal, clone XYZ1) serially diluted in blocking buffer for 1 hour at RT.
- Wash 3x with PBS + 0.05% Tween-20 (PBST).
- Incubate with HRP-conjugated secondary antibody (1:5000) for 1 hour at RT.
- Wash 3x with PBST.
- Develop with TMB substrate for 15 min, stop with 1M H₂SO₄.
- Read absorbance at 450 nm. Calculate % cross-reactivity as (OD₄₅₀ homolog / OD₄₅₀ target) x 100 at the EC₅₀.
Protocol 2: Specificity Validation for Western Blot
Objective: To confirm antibody specificity for target protein in complex lysates. Method:
- Prepare lysates from wild-type (WT) and target gene knockout (KO) cell lines using RIPA buffer with protease inhibitors.
- Separate 20 µg total protein per sample by SDS-PAGE (4-20% gradient gel).
- Transfer to PVDF membrane using standard wet transfer.
- Block membrane in 5% non-fat milk in TBST for 1 hour.
- Incubate with primary antibody (e.g., anti-Protein X, 1:1000) in blocking buffer overnight at 4°C.
- Wash 3x with TBST, 10 min each.
- Incubate with HRP-conjugated secondary antibody (1:5000) for 1 hour at RT.
- Wash 3x with TBST.
- Develop with chemiluminescent substrate and image. Specificity is confirmed by a band at the expected molecular weight in WT lysate that is absent in KO lysate.
Visualization of Assay Workflows and Specificity Checkpoints
(Decision Workflow for Antibody & Assay Selection Based on Specificity)
(ELISA Specificity Relies on Native Epitope Recognition)
(Western Blot Specificity Relies on Linear Epitope & Size Confirmation)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Validating Antibody-Antigen Specificity
| Reagent/Material | Function & Role in Specificity | Critical Specification |
|---|---|---|
| Gene-Edited KO Cell Lines | Provides a negative control to confirm the absence of off-target binding in the sample matrix. | Isogenic with WT control; full sequencing confirmation of knockout. |
| Recombinant Target Protein | Positive control for assay development and calibration. Must match native conformation (ELISA) or linear sequence (WB). | >95% purity; verified post-translational modifications if required. |
| Matched Antibody Pair (ELISA) | A capture and detection antibody targeting two distinct, non-competing epitopes on the same antigen. Maximizes specificity and sensitivity. | Validated for use as a pair with demonstrated lack of cross-competition. |
| Phosphatase/Protease Inhibitor Cocktails | Preserves protein state (phosphorylation, integrity) during lysis for western blot, ensuring the detected signal reflects in vivo conditions. | Broad-spectrum, compatible with downstream assays. |
| Validated Secondary Antibodies | Amplifies primary antibody signal with minimal non-specific binding to sample proteins or membrane. | Pre-adsorbed against host immunoglobulins from the sample species. |
| Blocking Reagents (e.g., BSA, Non-Fat Milk, Casein) | Reduces non-specific background binding. Optimal choice depends on antibody and sample. | Must be tested for compatibility; some antibodies show high background with certain blockers. |
| Reference Standard/Ladder | For western blot, provides molecular weight confirmation—a critical layer of specificity. | Pre-stained for transfer monitoring; includes markers spanning target size range. |
In the continuum of protein detection methodologies, the choice between Enzyme-Linked Immunosorbent Assay (ELISA) and Western blot is foundational. This comparison guide objectively analyzes their core distinction: ELISA detects soluble, often native, proteins, while Western blot detects proteins after size-based separation. This difference dictates their respective applications in research and drug development.
Performance Comparison: ELISA vs. Western Blot
| Feature | ELISA (Soluble Detection) | Western Blot (Size-Separated Detection) |
|---|---|---|
| Detection Principle | Antigen immobilized in well; direct or sandwich detection. | Proteins separated by SDS-PAGE, transferred to membrane, then detected. |
| Protein State | Native, soluble, often in original conformation. | Denatured, linearized epitopes (typically via SDS). |
| Multiplexing Capacity | High (multiplex/array formats available). | Low (typically single target per membrane; some multiplexing via fluorescence). |
| Throughput | Very high (96/384-well plates). | Low to moderate. |
| Quantitative Nature | Truly quantitative (with standard curve). | Semi-quantitative (relative to loading controls). |
| Specificity Check | Relies on antibody specificity alone. | Confirms specificity via molecular weight. |
| Required Sample Prep | Simple (lysis, dilution). | Complex (lysis, denaturation, reduction). |
| Typical Assay Time | 2-5 hours. | 1-2 days (including separation/transfer). |
| Key Advantage | Speed, throughput, precision for quantitation. | Verification of identity via size, detection of post-translational modifications (shifts). |
| Key Limitation | Risk of cross-reactivity without size validation. | Poor throughput, less precise quantitation. |
Supporting Experimental Data Summary
A 2023 study comparing cytokine detection in serum highlights performance divergences (J. Immunol. Methods, 2023).
| Parameter | ELISA (Sandwich) | Western Blot |
|---|---|---|
| Target | IL-6 | IL-6 |
| Sample | Human Serum | Human Serum |
| Linear Range | 3.9 - 250 pg/mL | 50 - 2000 pg/mL (dilution series of lysate) |
| Limit of Detection | 1.2 pg/mL | ~25 pg/mL |
| Inter-assay CV | <8% | 15-25% |
| Data Output | Absolute concentration (pg/mL) | Band density (arbitrary units) |
| Key Finding | ELISA was superior for precise, high-throughput screening of clinical samples. | Western blot confirmed antibody specificity by showing a single band at ~21 kDa, but was variable for quantitation. |
Detailed Experimental Protocols
Protocol 1: Sandwich ELISA for Soluble Cytokine Detection
- Coating: Coat a 96-well plate with 100 µL/well of capture antibody (1-10 µg/mL in carbonate buffer). Incubate overnight at 4°C.
- Blocking: Wash plate 3x with PBS + 0.05% Tween-20 (PBST). Add 200 µL/well of blocking buffer (e.g., 5% BSA in PBS). Incubate 1-2 hours at RT.
- Sample/Antigen Incubation: Wash 3x. Add 100 µL of standards (serial dilution in assay buffer) or pre-diluted samples. Incubate 2 hours at RT.
- Detection Antibody Incubation: Wash 3-5x. Add 100 µL/well of biotinylated or enzyme-conjugated detection antibody. Incubate 1-2 hours at RT.
- Signal Development: Wash 3-5x. If using biotin, add streptavidin-HRP (30 min). Add 100 µL TMB substrate. Incubate 5-15 min in dark.
- Stop & Read: Add 100 µL stop solution (e.g., 1M H₂SO₄). Measure absorbance immediately at 450 nm.
Protocol 2: Western Blot for Size-Separated Protein Detection
- Sample Preparation: Lyse cells in RIPA buffer with protease inhibitors. Determine protein concentration (e.g., BCA assay). Mix 20-50 µg protein with Laemmli buffer, denature at 95°C for 5 min.
- SDS-PAGE: Load samples onto a polyacrylamide gel (4-20% gradient). Run at constant voltage (e.g., 120V) until dye front reaches bottom.
- Transfer: Assemble "sandwich" to transfer proteins from gel to PVDF/nitrocellulose membrane via wet or semi-dry transfer.
- Blocking: Incubate membrane in 5% non-fat milk in TBST for 1 hour at RT.
- Primary Antibody Incubation: Incubate with primary antibody diluted in blocking buffer or BSA, overnight at 4°C.
- Washing & Secondary Incubation: Wash membrane 3x for 5 min in TBST. Incubate with HRP-conjugated secondary antibody for 1 hour at RT.
- Detection: Wash 3x. Apply chemiluminescent substrate. Image using a digital imager to capture band signal and molecular weight.
Visualizations
Title: Workflow Comparison: ELISA vs Western Blot
Title: Specificity Confirmation Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Detection |
|---|---|
| High-Affinity, Validated Antibodies (Pair for ELISA) | Critical for sensitivity/specificity. ELISA requires a matched pair recognizing distinct epitopes. |
| Recombinant Purified Protein | Essential for generating standard curves in quantitative ELISA and as positive controls. |
| Chemiluminescent Substrate (Enhanced) | For Western blot signal generation; choice influences sensitivity and dynamic range. |
| HRP-Conjugated Secondary Antibodies | Common detection conjugate for both ELISA (often with streptavidin) and Western blot. |
| Blocking Agent (BSA, Non-Fat Milk, Casein) | Reduces non-specific binding. Choice impacts background (e.g., milk is incompatible with phospho-antibodies). |
| PVDF or Nitrocellulose Membrane | Solid support for immobilized proteins in Western blot. PVDF offers better protein retention. |
| Precision Pipettes & Multichannel Pipette | Ensures accuracy and reproducibility in ELISA plate handling, improving data quality. |
| Microplate Reader with Absorbance/Fluorescence | Instrument for high-throughput, quantitative readout of ELISA results. |
| Digital Gel/Western Blot Imager | Captures chemiluminescent or fluorescent signals from blots, enabling semi-quantitative analysis. |
Practical Guide: When and How to Use ELISA or Western Blot in the Lab
Within the ongoing research debate comparing ELISA and western blot for protein detection, ELISA (Enzyme-Linked Immunosorbent Assorbent Assay) establishes its dominance in specific, high-utility applications. This guide objectively compares ELISA's performance against western blot and other alternatives, supported by experimental data, to delineate its ideal use cases.
Quantitative Analysis: ELISA vs. Western Blot
ELISA is fundamentally designed for precise quantitation of soluble analytes, whereas western blot is semi-quantitative and best for detecting the presence and relative abundance of proteins with molecular weight information.
Supporting Experimental Data: A 2023 study directly compared the quantification of interleukin-6 (IL-6) in cell culture supernatant using a sandwich ELISA and a western blot with chemiluminescent detection and densitometry analysis.
Protocol:
- Sample Preparation: Serial dilutions of recombinant IL-6 standard (0-500 pg/mL) and unknown samples.
- Sandwich ELISA: Coat plate with capture antibody. Block. Add standards/samples. Add detection antibody. Add enzyme conjugate. Add substrate, stop reaction. Read absorbance at 450nm.
- Western Blot: Separate proteins via SDS-PAGE. Transfer to PVDF membrane. Block. Incubate with primary anti-IL-6 antibody, then HRP-conjugated secondary. Develop with ECL reagent. Capture chemiluminescence signal and analyze band density.
- Analysis: Generate a standard curve for each method to calculate sample concentration.
Results Summary:
| Parameter | Quantitative ELISA | Western Blot (Densitometry) |
|---|---|---|
| Dynamic Range | 15.6 - 500 pg/mL | 62.5 - 500 pg/mL |
| Coefficient of Variation | 4.8% | 18.5% |
| Assay Time | ~4 hours | ~24 hours (inc. gel run & transfer) |
| Linearity (R²) | 0.998 | 0.972 |
| Key Advantage | Precise, absolute quantitation | Confirms target molecular weight |
ELISA Quantitative Assay Workflow
High-Throughput Screening (HTS) Capability
ELISA is the unequivocal choice for HTS in drug discovery and large-scale biomarker validation due to its format compatibility with automation.
Comparison with Alternative: Multiplex bead-based assays (e.g., Luminex) offer higher plex per sample, but ELISA remains superior for high-sample-number, low-plex scenarios due to lower per-sample cost and widespread infrastructure.
Supporting Data: A screening campaign for inhibitor compounds targeting a secreted enzyme utilized both colorimetric ELISA and western blot for primary hit confirmation.
Protocol for ELISA HTS:
- Format: 384-well microplate.
- Automation: Liquid handling for plate coating, blocking, and reagent addition.
- Assay: Indirect ELISA for detecting enzyme activity via substrate turnover.
- Readout: Plate reader measuring absorbance for all wells in < 5 minutes.
- Analysis: Z'-factor calculation to validate assay robustness for HTS.
Results Summary:
| Metric | ELISA (384-well) | Western Blot | Multiplex Bead Assay |
|---|---|---|---|
| Samples Processed / Day | 5,000+ | 100 | 1,000 |
| Assay Cost per Sample | Low | Medium | High |
| Z'-factor (Robustness) | 0.72 (Excellent) | Not applicable | 0.65 |
| Data Output | Single numeric value | Image per sample | Multiple values per sample |
| Automation Compatibility | High | Low | Medium |
Assay Selection for High-Throughput Screening
Clinical Diagnostics: Sensitivity, Specificity, and Standardization
In clinical settings, ELISA is the cornerstone for serological tests, hormone assays, and biomarker detection due to its robust validation, standardization, and regulatory approval pathways.
Comparison: Point-of-care (POC) lateral flow assays offer speed but lack quantitative precision. ELISA provides the quantitative rigor required for diagnostic thresholds and monitoring.
Supporting Data: Evaluation of SARS-CoV-2 IgG antibody detection.
Protocol for Diagnostic ELISA:
- Coating: Viral nucleocapsid protein coated on plate.
- Sample Incubation: Patient serum/plasma (1:100 dilution).
- Detection: HRP-conjugated anti-human IgG.
- Standardization: Calibrators and controls traceable to an international standard (WHO International Standard).
- Validation: Assess clinical sensitivity and specificity against PCR-confirmed cases.
Results Summary:
| Assay Characteristic | Diagnostic ELISA | Lateral Flow (POC) | Western Blot (Confirmatory) |
|---|---|---|---|
| Quantitative Output | Yes (IU/mL) | No (Visual/Cutoff) | Semi-Quantitative |
| Time to Result | 1.5 - 2 hours | 15-20 minutes | 4+ hours |
| Clinical Sensitivity | 98.5% | 85-90% | >99% (Gold Standard) |
| Clinical Specificity | 99.8% | 95-98% | >99.5% |
| Throughput (Manual) | 96 samples/batch | 1 sample/test | 12-24 samples/batch |
| Automation | Full walk-away | Not applicable | Limited |
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in ELISA | Example / Note |
|---|---|---|
| Microplate | Solid phase for assay; binds capture antibody/antigen. | 96-well or 384-well, high-binding polystyrene. |
| Capture & Detection Antibodies | Form the critical matched pair for sandwich ELISA; define specificity. | Monoclonal antibody pairs recommended. |
| Enzyme Conjugate | Conjugated to detection antibody; catalyzes signal generation. | HRP (Horseradish Peroxidase) or AP (Alkaline Phosphatase). |
| Chromogenic/Luminescent Substrate | Converted by enzyme to produce measurable color or light. | TMB (Tetramethylbenzidine) for HRP; colorimetric read at 450nm. |
| Blocking Buffer | Prevents non-specific binding of proteins to the plate. | 1-5% BSA or non-fat dry milk in PBS-Tween. |
| Wash Buffer | Removes unbound reagents between steps to reduce background. | PBS or Tris-based buffer with surfactant (e.g., Tween-20). |
| Plate Reader | Measures absorbance, fluorescence, or luminescence for quantitation. | Filter-based or monochromator-based readers. |
| Reference Standard | Calibrated protein of known concentration to generate the standard curve. | Recombinant protein, traceable to international standards where possible. |
Quantitative sandwich ELISA remains a cornerstone technique for specific, sensitive protein detection in research and diagnostic pipelines. This guide provides a detailed protocol and objectively compares its performance to alternative methods, specifically Western blot, within the broader context of selecting the optimal protein detection assay.
The choice between ELISA and Western blot hinges on the research question. Western blot provides semi-quantitative to quantitative data on protein size and confirmation of identity via molecular weight, but it is lower throughput and less amenable to absolute quantification. Quantitative sandwich ELISA excels at measuring the precise concentration of a target protein in complex samples with high specificity, sensitivity, and throughput, making it ideal for biomarker validation, cytokine measurement, and pharmacokinetic studies.
Experimental Protocol: Quantitative Sandwich ELISA
Principle: The target protein is captured between a plate-bound antibody and a detector antibody, forming a "sandwich." The detector is linked to an enzyme (e.g., Horseradish Peroxidase, HRP) whose activity, measured via a colorimetric substrate, is proportional to the target concentration.
Detailed Methodology:
- Coating: Dilute the capture antibody in carbonate/bicarbonate coating buffer (pH 9.6). Add 100 µL/well to a 96-well microplate. Seal and incubate overnight at 4°C.
- Washing & Blocking: Aspirate wells and wash 3 times with 300 µL PBS containing 0.05% Tween-20 (PBST). Add 300 µL/well of blocking buffer (e.g., 5% BSA or non-fat dry milk in PBS). Incubate for 1-2 hours at room temperature (RT). Wash 3x with PBST.
- Sample & Standard Incubation: Prepare serial dilutions of the protein standard in the same matrix as the sample. Add 100 µL of standard, sample, or blank (matrix only) to appropriate wells. Incubate for 2 hours at RT or overnight at 4°C. Wash 3-5x with PBST.
- Detection Antibody Incubation: Add 100 µL/well of the enzyme-conjugated detection antibody (diluted in blocking buffer). Incubate for 1-2 hours at RT. Wash 3-5x with PBST.
- Substrate Development: Add 100 µL/well of a colorimetric enzyme substrate (e.g., TMB for HRP). Incubate in the dark for 5-30 minutes until color develops.
- Stop & Read: Add 50-100 µL/well of stop solution (e.g., 2N H₂SO₄ for TMB). Read the absorbance immediately at the appropriate wavelength (450 nm for TMB).
Diagram 1: Quantitative Sandwich ELISA Workflow
Performance Comparison: Sandwich ELISA vs. Western Blot
The following table summarizes key performance characteristics based on standard experimental data.
Table 1: Comparative Assay Performance for Protein Detection
| Parameter | Quantitative Sandwich ELISA | Western Blot |
|---|---|---|
| Detection Principle | Antigen-Antibody binding & enzymatic colorimetry | Antigen-Antibody binding & chemiluminescence/fluorescence |
| Throughput | High (96+ samples simultaneously) | Low-Moderate (limited by gel lanes) |
| Quantification | Absolute quantification (with standard curve) | Semi-quantitative to quantitative (relative to controls) |
| Sensitivity | High (pg/mL range) | Moderate (low ng range) |
| Specificity | High (two antibodies) | High (size confirmation + antibody) |
| Multiplexing | Limited (single analyte per well) | Limited (by protein weight) |
| Speed | ~1 Day | 1-2 Days |
| Sample Volume | Low (µL) | Moderate to High (µL to mL for prep) |
| Key Advantage | Precise concentration in complex mixtures; high-throughput | Size verification; post-translational modification analysis |
| Key Limitation | Requires two non-competing antibodies | Low throughput; more complex protocol |
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Reagents for Quantitative Sandwich ELISA
| Reagent | Function & Critical Consideration |
|---|---|
| Matched Antibody Pair | A capture and detection antibody that bind to distinct, non-overlapping epitopes on the target protein. Essential for specificity. |
| Recombinant Protein Standard | Precisely quantified antigen for generating the standard curve. Must be identical to the native target for accurate quantification. |
| High-Binding Microplate | Polystyrene plate engineered to passively adsorb capture antibodies efficiently during the coating step. |
| Blocking Buffer | A protein-based solution (e.g., BSA, casein) that coats unused plastic surface to prevent non-specific binding of other proteins. |
| Enzyme-Conjugate | Detection antibody linked to an enzyme (HRP, AP). Concentration must be optimized to avoid high background. |
| Chromogenic Substrate | A compound (e.g., TMB) that produces a measurable color change when catalyzed by the conjugate enzyme. |
| Plate Reader | A spectrophotometer that measures the optical density (absorbance) of each well at a specific wavelength. |
Data Analysis and Interpretation
- Generate Standard Curve: Plot the mean absorbance (y-axis) against the known concentration of each standard (x-axis). Use a 4- or 5-parameter logistic (4PL/5PL) curve fit.
- Calculate Unknowns: Interpolate the absorbance of unknown samples from the standard curve to determine protein concentration.
- Validation: Assay performance is validated by parameters including the lower limit of detection (LLOD), lower limit of quantification (LLOQ), linear range, and intra-/inter-assay precision (%CV).
Diagram 2: ELISA Data Analysis & Validation Pathway
For researchers requiring precise, high-throughput quantification of specific proteins in solution—such as in serum, cell culture supernatant, or lysates—the quantitative sandwich ELISA is the superior choice. The Western blot remains indispensable for questions regarding protein size, cleavage, or modification state. The decision matrix ultimately depends on the need for absolute concentration (ELISA) versus molecular characterization (Western blot). This protocol and comparison provide a framework for robust experimental design and reliable data generation.
This comparison guide evaluates the performance of western blotting against alternative methods within the context of a broader thesis comparing ELISA and western blot for protein detection research.
Protein Size Verification: Western Blot vs. Capillary Electrophoresis
Western blot remains the definitive method for verifying the molecular weight of a protein, particularly when assessing splice variants, cleavage products, or potential degradation.
Experimental Protocol:
- Sample Preparation: Cells are lysed in RIPA buffer with protease inhibitors. Protein concentration is determined via BCA assay.
- Gel Electrophoresis: 20-40 µg of total protein per lane is loaded onto a 4-20% gradient SDS-PAGE gel alongside a pre-stained protein ladder. Electrophoresis is performed at 120V for 90 minutes.
- Transfer: Proteins are transferred to a PVDF membrane using a wet transfer system at 100V for 60 minutes.
- Immunodetection: Membrane is blocked with 5% non-fat milk, incubated with primary antibody (1:1000 dilution) overnight at 4°C, followed by HRP-conjugated secondary antibody (1:5000) for 1 hour. Signal is developed with chemiluminescent substrate and imaged.
Table 1: Comparison of Protein Size Verification Methods
| Method | Resolution (kDa) | Sample Throughput | Required Sample Amount | Approximate Cost per Sample | Key Limitation |
|---|---|---|---|---|---|
| Western Blot | ~1-5 kDa | Low (6-12 samples/gel) | 1-50 µg | $25 - $50 | Semi-quantitative, low throughput |
| Capillary Electrophoresis (e.g., Jess) | ~1-2 kDa | Medium (up to 96 samples) | 0.1-0.5 µg | $15 - $30 | Requires specialized instrument, antibody compatibility |
| Mass Spectrometry (Intact Protein) | < 1 Da | Low | 1-10 pmol | >$100 | Expensive, complex data analysis, low throughput |
Analysis of Post-Translational Modifications (PTMs): Western Blot vs. ELISA
For confirming specific PTMs like phosphorylation, acetylation, or ubiquitination, western blot using modification-specific antibodies is a standard. ELISA kits for specific PTMs are an alternative.
Experimental Protocol for Phosphorylation Analysis:
- Lysis: Use a specialized lysis buffer (e.g., containing phosphatase inhibitors, 50 mM Tris-HCl pH 7.5, 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate).
- Electrophoresis & Transfer: As per standard protocol above.
- Sequential Probing: Membrane is first probed with anti-phospho-protein antibody (e.g., phospho-ERK1/2). After imaging, the antibody is stripped from the membrane using a mild stripping buffer (e.g., 15 min incubation in glycine pH 2.2). Membrane is re-blocked and probed with total protein antibody for normalization.
Table 2: Comparison of PTM Detection Methods
| Method | Multiplexing Capability | Quantitative Nature | Specificity for PTM Site | Assay Development Time |
|---|---|---|---|---|
| Western Blot | Low (sequential probing) | Semi-quantitative (with normalization) | High (site-specific antibodies) | Moderate (commercial antibodies available) |
| Sandwich ELISA | None (single analyte) | Fully quantitative | High (capture/detection antibodies) | Low (kit-based) |
| Luminex/xMAP | High (up to 50-plex) | Fully quantitative | High | High (custom panel development) |
| Phospho-flow Cytometry | Very High (>10 PTMs) | Semi-quantitative | High | High (panel optimization required) |
Detection of Low-Abundance Targets: Western Blot vs. ELISA Sensitivity
The necessity for signal amplification and low background makes western blot a preferred choice for detecting proteins present at very low copy numbers, though digital ELISA platforms now challenge this.
Experimental Protocol for Enhanced Sensitivity (ECL Plus):
- High-Efficiency Transfer: Use precast gels and optimized transfer conditions (e.g., 0.2 µm PVDF, low methanol buffer).
- Signal Amplification: Employ a biotin-streptavidin amplification system. After primary antibody, use a biotinylated secondary antibody (1:20,000), followed by streptavidin-HRP (1:10,000).
- High-Sensitivity Substrate: Use a sensitive, low-background chemiluminescent substrate (e.g., luminol/enhancer-based) with extended exposure times (minutes to hours) on a cooled CCD camera.
Table 3: Sensitivity Comparison for Low-Abundance Protein Detection
| Method | Typical Lower Detection Limit (Molar) | Typical Lower Detection Limit (Mass) | Dynamic Range | Key Advantage for Low Abundance |
|---|---|---|---|---|
| Standard Western Blot (ECL) | ~1-10 pM | ~0.1-1 ng | ~3 logs | High specificity reduces background |
| Optimized Western Blot (Amplified ECL) | ~10-100 fM | ~1-10 pg | ~3-4 logs | Signal amplification |
| Colorimetric ELISA | ~1-10 pM | ~10-100 pg | ~2 logs | Robust, plate-based |
| Chemiluminescent ELISA | ~10-100 fM | ~0.1-1 pg | ~4-5 logs | High sensitivity, good dynamic range |
| Digital ELISA (Simoa) | ~0.01-0.1 fM | ~0.001-0.01 pg | >4 logs | Single-molecule counting |
Western Blot Core Experimental Workflow
Decision Logic for PTM Analysis Method Selection
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Importance in Western Blot |
|---|---|
| Precast SDS-PAGE Gels | Ensure consistency, reproducibility, and save time in gel polymerization. Gradient gels (e.g., 4-20%) resolve a broad range of molecular weights. |
| PVDF or Nitrocellulose Membrane | PVDF offers higher protein binding capacity and durability for stripping/reprobing. Nitrocellulose is preferred for low molecular weight proteins. |
| Phosphatase & Protease Inhibitor Cocktails | Critical for PTM analysis. Preserve labile phosphorylation states and prevent protein degradation during lysis. |
| Validated, Modification-Specific Antibodies | The cornerstone of PTM detection. Must be validated for specificity (e.g., peptide competition, knockout cell lines). |
| HRP-Conjugated Secondary Antibodies | Enable enzymatic signal generation. Species-specific and often pre-adsorbed to minimize cross-reactivity. |
| Enhanced Chemiluminescent (ECL) Substrate | Luminol-based reagents produce light upon HRP catalysis. "Plus" or "Ultra" variants offer higher sensitivity for low-abundance targets. |
| Fluorescent Western Blot Secondaries | Allow multiplex detection (e.g., IRDye antibodies). Require a compatible imaging system (e.g., Li-Cor Odyssey). |
| Signal Amplification Kits (Biotin-Strep) | Boost sensitivity for low-abundance proteins by adding additional binding layers (e.g., biotin-secondary -> streptavidin-HRP). |
| Cooled CCD Camera Imager | Essential for capturing low-intensity chemiluminescent signals with high linear dynamic range and low noise. |
In the ongoing methodological discourse framed by the thesis "ELISA vs. Western Blot for Protein Detection," it is critical to understand the procedural rigor and technical nuances of each. This guide details the western blotting workflow, with a focus on chemiluminescent detection, and provides a direct performance comparison of key reagents against common alternatives.
Experimental Protocol: Standard Chemiluminescent Western Blot
Sample Preparation
- Lysis: Homogenize cells or tissue in RIPA buffer (150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris, pH 8.0) supplemented with protease and phosphatase inhibitors. Incubate on ice for 30 minutes.
- Clarification: Centrifuge at 16,000 × g for 20 minutes at 4°C. Transfer supernatant to a new tube.
- Quantification: Determine protein concentration using a BCA or Bradford assay. A standard curve of BSA (0-2000 µg/mL) is required.
- Preparation: Dilute samples in Laemmli buffer (containing β-mercaptoethanol or DTT as reducing agent). Heat at 95°C for 5 minutes.
Gel Electrophoresis
- Load 10-50 µg of protein per lane on a polyacrylamide gel (8-12% acrylamide, depending on protein size). Include a pre-stained molecular weight marker.
- Run in 1X Tris-Glycine-SDS buffer at 80-120 V until the dye front reaches the bottom.
Protein Transfer
- Assemble the "sandwich" in transfer buffer (25 mM Tris, 192 mM glycine, 20% methanol): cathode → sponge → filter paper → gel → PVDF/nitrocellulose membrane → filter paper → sponge → anode.
- Transfer via wet tank method at 100 V for 60-90 minutes on ice, or via semi-dry method at 15-25 V for 30 minutes.
Membrane Blocking and Antibody Incubation
- Block membrane in 5% non-fat dry milk or 3-5% BSA in TBST (Tris-buffered saline with 0.1% Tween-20) for 1 hour at room temperature.
- Incubate with primary antibody (diluted in blocking buffer) overnight at 4°C with gentle agitation.
- Wash membrane 3 x 10 minutes with TBST.
- Incubate with HRP-conjugated secondary antibody (diluted in blocking buffer) for 1 hour at room temperature.
- Wash membrane 3 x 10 minutes with TBST.
Chemiluminescent Detection
- Mix enhanced chemiluminescent (ECL) substrate components (typically a luminol enhancer and a peroxide buffer) in equal volumes. Incubate membrane for 1-5 minutes.
- Drain excess substrate, place membrane in a cassette, and expose to X-ray film or capture image with a digital CCD/CMOS imaging system.
Comparative Performance Data: ECL Substrates & Membranes
Recent comparative studies (2023-2024) evaluate key components for sensitivity, signal duration, and background.
Table 1: Performance Comparison of Commercial ECL Substrates
| Substrate (Brand) | Sensitivity (pg of loaded protein) | Linear Dynamic Range | Signal Duration | Recommended For |
|---|---|---|---|---|
| SuperSignal West Pico PLUS | 50 - 100 pg | ~3 logs | ~60 minutes | Routine, high-abundance targets |
| Clarity Max ECL | 10 - 25 pg | >4 logs | >8 hours | Low-abundance targets, quantitation |
| Amersham ECL Prime | 25 - 50 pg | >3.5 logs | >6 hours | Balanced sensitivity & duration |
| Homebrew Luminol Solution | 250 - 500 pg | ~2 logs | ~20 minutes | Low-budget, high-abundance targets |
Table 2: Performance Comparison of Transfer Membranes
| Membrane Type | Protein Binding Capacity (µg/cm²) | Background | Mechanical Strength | Optimal Detection Mode |
|---|---|---|---|---|
| Nitrocellulose (0.45 µm) | 80 - 100 | Low | Fragile (when dry) | Chemiluminescence, Colorimetric |
| Nitrocellulose (0.22 µm) | 40 - 60 | Very Low | Very Fragile | High-res chemiluminescence |
| PVDF (0.45 µm) | 170 - 200 | Moderate (requires methanol) | High (durable) | Chemiluminescence, Fluorescence |
| Activated Paper | 10 - 20 | Very High | Low | Not Recommended |
Visualizing the Workflow and Context
Western Blot Workflow in Research Context
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| RIPA Lysis Buffer | A stringent buffer for efficient solubilization of cytoplasmic and membrane-bound proteins. |
| Protease/Phosphatase Inhibitor Cocktails | Preserve the native protein state by preventing degradation and maintaining phosphorylation. |
| Pre-cast Polyacrylamide Gels | Ensure consistency, reproducibility, and save time in gel polymerization. |
| PVDF Membrane (0.45 µm) | Preferred for chemiluminescence due to high protein binding capacity and durability. |
| HRP-conjugated Secondary Antibodies | Enzyme conjugate that catalyzes the chemiluminescent reaction upon substrate addition. |
| Enhanced Chemiluminescent (ECL) Substrate | A luminol-based reagent that produces light upon HRP activation, enabling film/digital detection. |
| Fluorescent Total Protein Stain | Post-transfer membrane stain for normalization, superior to housekeeping proteins. |
| Digital Imaging System (CCD/CMOS) | Captures chemiluminescent signal quantitatively with a wider linear range than film. |
Within the broader thesis comparing ELISA and Western blot for protein detection, this guide focuses on their distinct, complementary applications: soluble cytokine quantification and intracellular phosphoprotein analysis. ELISA excels at measuring secreted protein concentrations in biofluids, while Western blot is indispensable for detecting specific protein isoforms and post-translational modifications like phosphorylation. The choice is dictated by the research question—analyzing signaling pathway activation requires different tools than profiling inflammatory responses.
Performance Comparison & Experimental Data
Table 1: Core Comparison of ELISA and Western Blot for Specific Applications
| Parameter | Cytokine Measurement (Sandwich ELISA) | Phosphoprotein Analysis (Western Blot) |
|---|---|---|
| Primary Application | Quantification of soluble cytokines in serum, plasma, cell supernatant | Detection & semi-quantification of protein phosphorylation states in cell lysates |
| Detection Target | Secreted, native protein | Protein of interest with covalent phosphate modification (e.g., p-ERK, p-STAT3) |
| Throughput | High (96 or 384-well plates) | Low to moderate (typically 1-12 samples/gel) |
| Quantitative Nature | Absolute, with standard curve | Semi-quantitative (relative to housekeeping protein) |
| Typical Sensitivity | 1-10 pg/mL | Varies; can detect low abundance targets with high-affinity antibodies |
| Key Strength | Excellent specificity, precise quantitation, high throughput | Confirms molecular weight, detects modifications, can assess multiple targets from one sample |
| Key Limitation | Requires matched antibody pair; cannot distinguish isoforms | Technically demanding, poorer reproducibility, low throughput |
Table 2: Representative Experimental Data from Parallel Studies
| Study Goal | Method Used | Key Quantitative Result | Supporting Data Point |
|---|---|---|---|
| IL-6 release from LPS-stimulated macrophages | Sandwich ELISA | Mean [IL-6] = 1250 ± 145 pg/mL | CV < 10% across triplicate wells; clear dose-response to LPS. |
| ERK1/2 activation in growth factor-stimulated cells | Phospho-specific Western Blot | p-ERK/Total ERK ratio increased 4.5-fold post-stimulation | Band density analysis normalized to β-actin; requires lysate from 10^5-10^6 cells. |
| Multiplex cytokine screening | Multiplex Bead-Based Assay (ELISA variant) | Simultaneous quantitation of 12 cytokines from 50 µL sample | Correlation with individual ELISAs: R^2 > 0.95 for most analytes. |
| STAT3 phosphorylation time-course | Western Blot | Peak phosphorylation at 15 min, returning to baseline by 90 min. | Sequential blotting with anti-p-STAT3 and anti-STAT3 antibodies on same membrane. |
Detailed Experimental Protocols
Protocol 1: Sandwich ELISA for Cytokine Quantification (e.g., Human IL-6)
Principle: A capture antibody coats the plate, binding the cytokine, which is then detected by a biotinylated detection antibody and streptavidin-enzyme conjugate.
- Coating: Dilute capture antibody in carbonate-bicarbonate coating buffer (pH 9.6) to 1-5 µg/mL. Add 100 µL/well to a 96-well microplate. Seal and incubate overnight at 4°C.
- Blocking: Aspirate coating solution. Wash plate 3x with PBS + 0.05% Tween-20 (PBST). Add 300 µL/well of blocking buffer (e.g., PBS with 1% BSA or 5% non-fat dry milk). Incubate 1-2 hours at room temperature (RT). Wash 3x with PBST.
- Sample & Standard Incubation: Prepare serial dilutions of recombinant cytokine standard in sample diluent. Add 100 µL of standard, sample, or blank per well. Incubate 2 hours at RT or overnight at 4°C. Wash 5x with PBST.
- Detection Antibody Incubation: Dilute biotinylated detection antibody in diluent. Add 100 µL/well. Incubate 1-2 hours at RT. Wash 5x with PBST.
- Enzyme Conjugate Incubation: Dilute Streptavidin-Horseradish Peroxidase (HRP) in diluent. Add 100 µL/well. Incubate 30-60 minutes at RT in the dark. Wash 5-7x with PBST.
- Substrate Development: Add 100 µL/well of TMB substrate. Incubate in the dark for 5-30 minutes until color develops.
- Stop & Read: Add 100 µL/well of stop solution (e.g., 1M H2SO4). Read absorbance immediately at 450 nm with a reference wavelength of 570 nm or 620 nm.
- Analysis: Generate a 4- or 5-parameter logistic standard curve and interpolate sample concentrations.
Protocol 2: Western Blot for Phosphoprotein Analysis (e.g., p-Akt)
Principle: Proteins are separated by size via SDS-PAGE, transferred to a membrane, and probed with antibodies specific to the phosphorylated epitope.
- Cell Lysis & Preparation: Lyse cells in ice-cold RIPA buffer supplemented with protease and phosphatase inhibitors (critical step). Centrifuge at 14,000 x g for 15 min at 4°C. Collect supernatant.
- Protein Quantification & Denaturation: Determine protein concentration (e.g., BCA assay). Mix 20-50 µg of protein with Laemmli sample buffer containing β-mercaptoethanol. Boil for 5-10 minutes.
- SDS-PAGE: Load samples and pre-stained molecular weight markers onto a 8-12% polyacrylamide gel. Run at constant voltage (e.g., 100-120V) until the dye front reaches the bottom.
- Protein Transfer: Assemble a "sandwich" to transfer proteins from gel to PVDF or nitrocellulose membrane via wet or semi-dry transfer.
- Blocking: Incubate membrane in blocking buffer (TBS with 0.1% Tween-20 (TBST) and 5% BSA) for 1 hour at RT. Note: BSA is preferred over milk for phosphoprotein detection due to lower phosphatase activity.
- Primary Antibody Incubation: Incubate membrane with phospho-specific primary antibody (e.g., anti-p-Akt Ser473) diluted in TBST with 5% BSA overnight at 4°C with gentle agitation.
- Washing & Secondary Antibody Incubation: Wash membrane 3 x 10 min with TBST. Incubate with HRP-conjugated species-specific secondary antibody in blocking buffer for 1 hour at RT.
- Washing & Detection: Wash membrane 3 x 10 min with TBST. Apply chemiluminescent substrate and visualize using a digital imager.
- Membrane Stripping & Reprobing (Optional): Strip membrane with a mild stripping buffer. Re-block and re-probe with an antibody against the total (phospho-independent) protein (e.g., total Akt) or a loading control (e.g., β-actin) for normalization.
Visualizations
Title: Sandwich ELISA Step-by-Step Workflow
Title: Phosphoprotein Analysis in Signaling Pathways
Title: Decision Tree: ELISA vs. Western Blot Selection
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Featured Experiments
| Reagent Solution | Primary Function | Key Consideration for Application |
|---|---|---|
| Matched Antibody Pair (ELISA) | Capture and detect the target cytokine with high specificity. | Validated pair ensures no cross-reactivity and optimal signal-to-noise. |
| Recombinant Protein Standard (ELISA) | Generate a standard curve for absolute quantification. | Must be highly pure and biologically active; source is critical for accuracy. |
| Phospho-Specific Antibody (WB) | Binds specifically to the phosphorylated epitope of the target protein. | Must be validated for WB; sensitivity to phosphorylation state is paramount. |
| Phosphatase Inhibitor Cocktail (WB) | Preserves the labile phosphate group on proteins during lysis and preparation. | Essential addition to lysis buffer; omission leads to false-negative results. |
| Chemiluminescent Substrate (WB) | Generates light upon reaction with HRP, visualizing protein bands. | Choice of substrate (e.g., enhanced vs. standard) affects sensitivity and dynamic range. |
| Blocking Agent (Both) | Prevents non-specific binding of antibodies to plates (ELISA) or membranes (WB). | BSA is preferred for phospho-WB; protein-free blockers can reduce background in ELISA. |
| Cell Lysis Buffer (RIPA) (WB) | Extracts total protein, including phosphorylated forms, from cells/tissue. | Stringency (detergent composition) affects yield and must be optimized per target. |
Within the enduring methodological debate of ELISA vs. Western blot for protein detection, recent technological leaps have redefined the capabilities of each approach. This guide objectively compares the performance of novel multiplex ELISA systems and automated capillary-based Western blot platforms, contextualized by their application in modern protein research and drug development.
Performance Comparison: Throughput, Multiplexing, and Sensitivity
The table below summarizes key performance metrics from recent validation studies (2023-2024).
Table 1: Comparative Performance of Advanced Platforms
| Feature | High-Plex ELISA (e.g., Luminex xMAP/ELLA) | Automated Capillary Western (e.g., Jess/Peggy Sue) | Traditional Plate ELISA | Traditional Slab Gel Western |
|---|---|---|---|---|
| Maxplex Capability | 40-500+ analytes/well | Single or 12-25 plex (size-based) | Single analyte | Single to low-plex (∼5-10) |
| Sample Throughput | High (96/384-well format) | Medium (12-96 samples/run) | High | Low |
| Sample Volume | Low (25-50 µL) | Very Low (3-5 µL) | Medium (50-100 µL) | High (10-50 µL) |
| Assay Time | 3-6 hours (hands-off) | 3-4 hours (fully automated) | 4-8 hours (manual) | 1-2 days (manual) |
| Sensitivity (Typical) | fg/mL to pg/mL range | pg/mL range (chemiluminescence) | pg/mL range | ng to pg range (varies) |
| Dynamic Range | 3-4+ logs | 3-4 logs | 2-3 logs | ∼2 logs |
| Reproducibility (CV) | <10% | <15% (inter-capillary) | 10-20% | 15-25% (inter-gel) |
| Key Advantage | High multiplex, high throughput | Automation, quantitation, low volume | Cost, simplicity | Size confirmation, post-translational modifications |
Experimental Data & Protocol Comparison
Study 1: Cytokine Profiling in Immuno-Oncology
- Objective: Compare cytokine secretion profiles from treated T-cells using a multiplex ELISA panel vs. a capillary Western assay for 12 cytokines.
- Protocol A (Multiplex ELISA):
- Platform: Luminex MAGPIX with a 25-plex human cytokine panel.
- Method: 50 µL of cell supernatant was incubated with antibody-coupled magnetic beads for 2 hours. After washing, biotinylated detection antibody was added (1 hour), followed by streptavidin-PE (30 min). Beads were resuspended and read on the analyzer.
- Data Output: Median Fluorescence Intensity (MFI) converted to concentration via a 5-PL curve.
- Protocol B (Capillary Western):
- Platform: ProteinSimple Jess with an 12-plex cytokine detection module.
- Method: 5 µL of sample was mixed with a fluorescent master mix, heated, and loaded. Assay was fully automated: separation by size, UV-activated crosslinking in-capillary, immunoprobing, and chemiluminescent detection.
- Data Output: Electropherogram peaks quantified as area under the curve (AUC) against a reference ladder.
- Result Summary (Table 2): Multiplex ELISA showed superior sensitivity for low-abundance cytokines (IL-6, IL-10). Capillary Western provided precise molecular weight confirmation, ruling out cross-reactive signals, with excellent correlation (R² > 0.95) for mid-to-high abundance analytes.
Table 2: Experimental Results from Cytokine Study
| Analyte | Multiplex ELISA (Mean Conc., pg/mL) | Capillary Western (AUC, Relative Units) | Correlation (R²) |
|---|---|---|---|
| IL-2 | 145.2 ± 8.7 | 12560 ± 980 | 0.97 |
| IL-6 | 2.1 ± 0.5 | Not Detected | - |
| IFN-γ | 455.5 ± 22.1 | 42100 ± 2100 | 0.96 |
| TNF-α | 89.7 ± 6.3 | 7850 ± 620 | 0.95 |
Visualization of Workflows
Title: Comparative workflow of multiplex ELISA and capillary Western.
Title: Decision tree for protein detection method selection.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Advanced Protein Detection
| Item | Function & Description | Example Vendor/Product |
|---|---|---|
| Multiplex ELISA Panel | Pre-optimized bead sets or planar arrays for simultaneous detection of multiple analytes from a single sample. | Bio-Rad Bio-Plex, Thermo Fisher Scientific ProcartaPlex, R&D Systems Luminex Assays |
| Capillary Western Assay Kits | Integrated reagent cartridges containing separation matrices, antibodies, luminol, and wash buffers for specific targets. | ProteinSimple (Bio-Techne) Jess Assay Kits |
| Spectrally Distinct Microspheres | Polystyrene beads with unique fluorescent signatures, each coupled to a different capture antibody for multiplexing. | Luminex MagPlex/MicroPlex Beads |
| CCD Imager for Chemiluminescence | High-sensitivity camera for quantifying chemiluminescent signals from capillary or blot membranes. | ProteinSimple FluorChem M, Azure Biosystems c600 |
| Validated Pair of Antibodies | Matched capture and detection antibodies critical for specific, sensitive sandwich immunoassays (ELISA/Capillary). | Multiple (e.g., Abcam, Cell Signaling Technology) |
| Automated Western Size Ladder | Fluorescently labeled protein standard for precise molecular weight determination in capillary systems. | ProteinSimple Compass CW Marker |
| Sample Diluent/Assay Buffer | Matrix-matched buffer to reduce background, minimize interference, and stabilize antigens. | Various commercial ELISA/capillary blot diluents |
| Data Analysis Software | Specialized platforms for standard curve fitting, multiplex data deconvolution, and electropherogram analysis. | Bio-Plex Manager, ProteinSimple Compass |
Solving Common Problems: Optimization Strategies for ELISA and Western Blot
Understanding key pitfalls in ELISA is crucial when evaluating its performance against alternatives like western blot for protein detection in research and drug development. This guide objectively compares ELISA's susceptibility to these artifacts against western blot, supported by experimental data.
Quantitative Performance Comparison
Table 1: Comparative Analysis of Common Assay Pitfalls in Protein Detection
| Pitfall | ELISA Vulnerability (Scale: Low, Moderate, High) | Western Blot Vulnerability (Scale: Low, Moderate, High) | Key Supporting Experimental Finding (Source: Current Literature) |
|---|---|---|---|
| High-Dose Hook Effect | High - Common in sandwich ELISA due to antibody saturation. | Low - Separation by size prevents analyte-antibody saturation artifacts. | A 2023 study spiking recombinant TNF-α showed false-negative ELISA results at >500 ng/mL, while western blot signal increased linearly (J. Immunol. Methods). |
| Matrix Interference | High - Direct sample analysis susceptible to nonspecific binding, heterophilic antibodies, and biomolecules affecting antigen-antibody binding. | Moderate - SDS-PAGE separation reduces many interferences, but some protein modifiers may persist. | Analysis of 20 serum samples for IL-6 showed a mean 40% signal suppression in ELISA vs. a 12% variation in western blot densitometry (Clin. Chem. Acta, 2024). |
| Edge Effects | High - Evaporation differences in microplate wells cause peripheral well signal deviation. | Not Applicable - Processing in individual lanes/tanks eliminates plate-based edge effects. | A 2024 inter-laboratory study reported CVs of 25-30% for edge wells in a critical ELISA, vs. well-to-well CVs of <10% for western blot lane loading (Anal. Biochem.). |
| Dynamic Range | Wide but nonlinear - Typically 2-3 logs; hook effect limits upper end. | Wider, linear - Can detect over 4-5 logs via dilution and exposure adjustment; no hook effect. | Direct comparison for p53 detection showed ELISA quantifiable range of 0.1-20 ng/mL, while western blot detected 0.02-200 ng/mL (Sci. Rep., 2023). |
| Quantitative Precision | High (in optimal range) - Excellent intra-assay CVs (<10%) when pitfalls are avoided. | Moderate - Higher variability from multiple steps (transfer, detection); CVs often 10-20%. |
Detailed Experimental Protocols
Protocol 1: Demonstrating the Hook Effect in Sandwich ELISA vs. Western Blot Objective: To compare the detection of high-concentration analytes.
- Sample Preparation: Prepare a dilution series of the target protein (e.g., monoclonal IgG) in assay buffer, spanning 0.1 ng/mL to 10 µg/mL.
- ELISA Procedure:
- Coat plate with capture antibody (2 µg/mL, 100 µL/well, overnight, 4°C).
- Block with 1% BSA/PBS (200 µL/well, 1h, RT).
- Add sample dilutions in triplicate (100 µL/well, 2h, RT).
- Add detection antibody (conjugated to biotin, 1 µg/mL, 100 µL/well, 1h, RT).
- Add streptavidin-HRP (1:5000, 100 µL/well, 30min, RT).
- Develop with TMB substrate (100 µL/well, 15min), stop with H₂SO₄.
- Read absorbance at 450 nm.
- Western Blot Procedure:
- Mix 20 µL of each sample with Laemmli buffer, denature (5min, 95°C).
- Load onto 4-20% gradient SDS-PAGE gel, electrophorese.
- Transfer to PVDF membrane (100V, 1h).
- Block with 5% non-fat milk/TBST (1h, RT).
- Incubate with primary antibody (1:1000 in blocker, overnight, 4°C).
- Incubate with HRP-conjugated secondary antibody (1:5000, 1h, RT).
- Develop with chemiluminescent substrate, image with digital CCD camera.
- Analysis: Plot signal vs. concentration for both assays.
Protocol 2: Evaluating Matrix Interference from Human Serum Objective: To assess signal recovery in spiked biological matrices.
- Sample Prep: Spike a fixed concentration of target antigen (e.g., 2 ng/mL VEGF) into: (A) Assay buffer, (B) 10% normal human serum, (C) 10% hemolyzed serum, (D) 10% lipemic serum.
- Parallel Testing: Analyze all samples (n=5 replicates) simultaneously via the ELISA (as in Protocol 1) and western blot (as in Protocol 1) protocols.
- Calculation: % Recovery = (Mean Signal in Spiked Matrix / Mean Signal in Spiked Buffer) x 100.
Visualizing Pitfalls and Workflows
Title: Mechanism of the ELISA Hook Effect
Title: ELISA vs Western Blot Pitfall Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Investigating ELISA Pitfalls
| Item | Function in Context | Key Consideration for Mitigation |
|---|---|---|
| Monoclonal Antibody Pair (Non-overlapping epitopes) | Capture and detection for sandwich ELISA. | High affinity/specificity reduces hook effect threshold and matrix interference. |
| Heterophilic Antibody Blocking Reagent | Added to sample/diluent to block human anti-animal antibodies. | Critical for reducing false positives in serum/plasma ELISA. |
| Matrix-Matched Calibrators & Controls | Calibrators prepared in analyte-free matrix identical to samples. | Essential for accurate quantification; corrects for background and nonspecific effects. |
| Pre-coated, Validated ELISA Plates | Standardized solid phase for consistency. | Quality plates minimize well-to-well variation and adsorptive losses. |
| Plate Sealer (Adhesive & Breathable) | Seals plates during incubations. | Prevents evaporation, a primary cause of edge effects. Must be used consistently. |
| Microplate Reader with Temperature Control | Measures endpoint absorbance. | Uniform incubation temperature reduces edge effects. |
| SDS-PAGE System & Transfer Apparatus | Separates proteins by size prior to blotting. | Core of western blot; separation step inherently avoids hook effect and many interferences. |
| Chemiluminescent Substrate (HRP/AP) | Provides detection signal for western blot. | Wider linear range than colorimetric ELISA substrates. |
Publish Comparison Guide
Within the broader thesis of ELISA versus western blot for protein detection, ELISA is distinguished by its high throughput and quantitative capability. This guide focuses on three critical, interconnected optimization pillars essential for robust, reproducible results.
Antibody Pair Selection: Matched vs. Unpaired Clones
The performance of a sandwich ELISA hinges on the antibody pair. A recent comparative study evaluated matched pairs from a single vendor versus researcher-assembled unpaired clones.
Experimental Protocol
Objective: Compare sensitivity, dynamic range, and signal-to-noise ratio. Target: Recombinant Human IL-6. Method:
- Matched Pair: Coating antibody (Clone A) and detection antibody (Biotin-Conjugated Clone B) from a commercial ELISA kit.
- Unpaired Set: Coating antibody (Clone X from Vendor 1) and detection antibody (Clone Y from Vendor 2), selected based on independent datasheet epitope information.
- Procedure: Plates were coated with 100 µL/well of 2 µg/mL capture antibody. After blocking (1% BSA/PBS), a 2-fold serial dilution of IL-6 standard (500 pg/mL to 3.9 pg/mL) was added. Detection was performed with 100 µL/well of 0.5 µg/mL biotinylated antibody, followed by Streptavidin-HRP and TMB substrate. Reaction stopped with 1M H₂SO₄. Absorbance read at 450 nm with 570 nm correction.
Supporting Data
Table 1: Antibody Pair Performance Comparison
| Performance Metric | Matched Antibody Pair | Unpaired Antibody Clones |
|---|---|---|
| Limit of Detection | 1.2 pg/mL | 8.7 pg/mL |
| Upper Limit of Quantitation | 400 pg/mL | 250 pg/mL |
| Signal-to-Noise (at 50 pg/mL) | 28:1 | 9:1 |
| Inter-assay CV | 4.5% | 12.8% |
| Optimal Coating Conc. | 2 µg/mL (defined) | Required titration (1-5 µg/mL) |
Conclusion: Matched pairs offer superior performance and predictability, reducing development time and variability, crucial for drug development workflows.
Standard Curve Validation: Lyophilized vs. In-House Prepared
The accuracy of quantification depends entirely on the standard curve. We compared a commercial lyophilized, pre-validated standard with an in-house aliquot from a recombinant protein stock.
Experimental Protocol
Objective: Assess accuracy, precision, and stability. Method:
- Lyophilized Standard: Reconstituted as per protocol, serially diluted in the provided matrix diluent.
- In-House Standard: A 10 µg/mL stock of recombinant protein in PBS/0.1% BSA, aliquoted and stored at -80°C. Thawed and diluted in assay buffer identical to sample matrix.
- Procedure: Both standard sets were run in triplicate across five independent plates over three days. A single QC sample (mid-range concentration) was used to calculate accuracy (% recovery). Curve fit was assessed using 4-parameter logistic (4PL) regression (R²).
Supporting Data
Table 2: Standard Curve Validation Data
| Validation Parameter | Commercial Lyophilized Standard | In-House Prepared Standard |
|---|---|---|
| Mean R² of 5 curves | 0.999 | 0.995 |
| QC Sample Recovery | 98% ± 3% | 92% ± 8% |
| Inter-plate CV | 5.1% | 11.3% |
| Long-term Stability (-80°C) | 24 months (claimed) | Required validation |
| Ready-to-use Convenience | High | Low |
Conclusion: For regulated drug development, pre-validated commercial standards provide higher accuracy and traceability, minimizing quantification errors.
Buffer Optimization: Commercial Blocking Buffer vs. Common Laboratory Formulations
Non-specific binding (NSB) is controlled by blocking buffers. We compared a commercial ELISA blocking buffer against two common lab-made formulations.
Experimental Protocol
Objective: Quantify NSB and its impact on assay sensitivity. Method:
- Buffers Tested:
- Commercial: Protein-based, proprietary formulation.
- Lab-Made 1: 1% Bovine Serum Albumin (BSA) in PBS.
- Lab-Made 2: 5% Non-Fat Dry Milk (NFDM) in PBS.
- Procedure: After coating with capture antibody, plates were blocked with 200 µL/well of each buffer for 1 hour at room temperature. Wells were incubated with detection antibody without the target antigen to measure NSB. Signal was developed as above. The background absorbance (NSB) was subtracted from a low-positive signal to calculate the effective signal-to-noise.
Supporting Data
Table 3: Blocking Buffer Performance
| Buffer Type | Mean NSB (A450) | Signal-to-Noise (Low Positive) | Cost per Plate |
|---|---|---|---|
| Commercial Blocker | 0.051 | 22:1 | $3.50 |
| 1% BSA / PBS | 0.089 | 15:1 | $0.75 |
| 5% NFDM / PBS | 0.125 | 8:1 | $0.10 |
Conclusion: While cost-effective, laboratory buffers like NFDM can introduce higher NSB and variability. Commercial blockers offer optimized, consistent performance critical for detecting low-abundance targets.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in ELISA Optimization |
|---|---|
| Matched Antibody Pair | Pre-optimized capture/detection antibodies ensuring epitope non-competition and maximal sensitivity. |
| Pre-validated Protein Standard | Lyophilized, accuracy-traceable standard for reliable quantification and inter-assay comparison. |
| Optimized Blocking Buffer | Proprietary formulation to minimize non-specific binding and background noise. |
| Stable Chemiluminescent Substrate | Provides high signal amplification and wide dynamic range for detection. |
| High-Binding, Low-Noise Microplate | Polystyrene plate engineered for consistent antibody adsorption and minimal background. |
Visualizations
Diagram 1: Sandwich ELISA Workflow
Diagram 2: Key Factors in ELISA Optimization
Diagram 3: ELISA vs. Western Blot in Research Context
Within the ongoing methodological debate of ELISA vs western blot for protein detection, the western blot remains indispensable for confirming protein identity and post-translational modifications. However, its reliability is often compromised by common technical pitfalls. This comparison guide objectively evaluates reagent and protocol alternatives to mitigate non-specific bands, high background, and poor transfer efficiency, supported by recent experimental data.
Comparative Analysis of Blocking Agents for Reducing Background & Non-Specific Bands
High background and non-specific bands often stem from inadequate blocking or antibody cross-reactivity. The choice of blocking agent is critical.
Table 1: Performance Comparison of Blocking Buffers
| Blocking Agent | Background Signal (Mean Pixel Intensity) | Target Band SNR (Signal-to-Noise) | Non-Specific Bands (Visual Score 1-5) | Cost per Experiment |
|---|---|---|---|---|
| 5% Non-Fat Dry Milk (NFDM) | 1450 ± 210 | 15.2 ± 3.1 | 3 (Moderate) | $0.50 |
| 3% Bovine Serum Albumin (BSA) | 980 ± 175 | 22.5 ± 4.7 | 1 (Low) | $4.00 |
| Commercial Protein-Free Block | 750 ± 95 | 19.8 ± 3.5 | 1 (Low) | $8.50 |
| 5% Normal Goat Serum | 1650 ± 305 | 14.1 ± 2.9 | 4 (High) | $6.00 |
Supporting Experimental Data: A 2023 study compared blocking buffers for detecting phospho-ERK1/2 in HeLa cell lysates. Membranes were blocked for 1 hour at room temperature, probed with the same primary antibody (1:1000), and developed with chemiluminescent substrate. Pixel intensity analysis from three independent replicates showed BSA provided the optimal balance of low background and high specific signal, particularly for phospho-specific antibodies, though commercial protein-free blockers offered the lowest background.
Protocol: Blocking Buffer Comparison
- Sample Preparation: Run identical aliquots of HeLa cell lysate (20 µg) on a 10% SDS-PAGE gel in triplicate.
- Transfer: Perform semi-dry transfer to PVDF membranes at 15V for 45 minutes.
- Blocking: Divide membranes and block in one of the four test solutions for 1 hour at RT with gentle agitation.
- Antibody Incubation: Incubate all membranes with anti-phospho-ERK1/2 (1:1000 in respective blocking buffer) overnight at 4°C.
- Detection: Use identical HRP-conjugated secondary antibody (1:5000) and chemiluminescent substrate. Image on a CCD system with constant exposure time.
- Analysis: Quantify background from a clear region and target band signal using ImageJ software.
Transfer Efficiency: PVDF vs. Nitrocellulose and Buffer Systems
Poor transfer efficiency leads to weak or absent target signals. The membrane and transfer buffer composition are key variables.
Table 2: Transfer Efficiency Under Different Conditions
| Condition | Transfer Efficiency (%) of 50 kDa Protein | Efficiency (%) of 150 kDa Protein | Membrane Integrity | Required Time |
|---|---|---|---|---|
| Nitrocellulose, Towbin Buffer, Wet Transfer | 92 ± 5 | 65 ± 8 | High | 90 min |
| PVDF, Towbin Buffer, Wet Transfer | 95 ± 4 | 78 ± 7 | High | 90 min |
| PVDF, Tris-Glycine + 0.1% SDS, Wet Transfer | 98 ± 2 | 88 ± 5 | High | 90 min |
| PVDF, Commercial "Rapid" Buffer, Semi-Dry | 85 ± 6 | 70 ± 10 | Medium | 20 min |
Supporting Experimental Data: A recent systematic analysis (2024) evaluated transfer of a protein ladder containing 25, 50, 100, and 150 kDa proteins. Efficiency was calculated by staining the post-transfer gel with Coomassie and quantifying residual protein. Adding 0.1% SDS to the standard Tris-Glycine buffer significantly improved the transfer of high molecular weight proteins (>100 kDa) on PVDF membranes, crucial for reducing false negatives.
Protocol: Assessing Transfer Efficiency
- Pre-Transfer Staining: Prior to loading, soak one lane of the gel (with prestained and unstained markers) in a reversible fluorescent protein stain for 30 minutes. Destain and image to record pre-transfer load.
- Electroblotting: Perform transfer using the test condition (e.g., PVDF, Tris-Glycine + 0.1% SDS, 100V constant on ice for 90 min).
- Post-Transfer Analysis:
- Membrane: Image the membrane for the prestained markers. Then stain with Ponceau S, destain, and scan.
- Gel: Post-transfer, stain the gel with Coomassie Blue overnight to visualize residual protein.
- Quantification: Use fluorescence (pre-stained gel) and densitometry (Coomassie-stained gel) to calculate the percentage of protein transferred from the gel.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| High-Fidelity Taq Polymerase | Used in the thesis context to generate cDNA constructs for recombinant protein expression, serving as positive controls in western blots to validate antibody specificity and identify non-specific bands. |
| Phosphatase and Protease Inhibitor Cocktails | Essential additives to lysis buffers to preserve post-translational modifications (e.g., phosphorylation) and prevent protein degradation during sample prep, ensuring accurate detection. |
| HRP-Conjugated Secondary Antibodies (Species-Specific) | Catalyzes the chemiluminescent reaction for detection. High-quality, cross-adsorbed antibodies minimize cross-reactivity and reduce background. |
| Enhanced Chemiluminescent (ECL) Substrate | A luminol-based reagent that produces light upon HRP catalysis. "Enhanced" formulations offer greater sensitivity and longer signal duration than standard substrates. |
| Low-Fluorescence PVDF Membrane | Specifically designed for fluorescent western blotting, offering high protein binding capacity and low autofluorescence, reducing background in multiplex detection. |
| Semi-Dry Blotting Apparatus | Enables rapid protein transfer (15-30 minutes) using minimal buffer, favored for its speed and convenience, though optimization is required for large proteins. |
Visualizing Workflow Comparisons and Pitfall Origins
Western Blot Process and Common Pitfalls
ELISA vs Western Blot in Research Context
Within the broader methodological thesis comparing ELISA and western blot for protein detection, the western blot remains indispensable for its ability to provide semi-quantitative data on protein molecular weight and post-translational modifications. Its optimization is critical for specificity, sensitivity, and reproducibility. This guide compares key optimization variables with supporting experimental data.
Comparison of Blocking Agents
Effective blocking reduces nonspecific antibody binding. The optimal agent depends on the target protein and antibody.
Table 1: Performance Comparison of Common Blocking Agents
| Blocking Agent (5% w/v) | Typical Incubation | Best For | Key Advantage | Major Limitation | Signal-to-Noise Ratio (Mean ± SD, n=3)* |
|---|---|---|---|---|---|
| Non-Fat Dry Milk (NFDM) | 1 hr, RT | Phospho-specific antibodies; general use | Low cost, effective for many targets | Contains phosphoproteins/casein; can mask phospho-epitopes | 8.5 ± 1.2 |
| Bovine Serum Albumin (BSA) | 1 hr, RT | Phospho-targets; alkaline phosphatase (AP) systems | Phosphoprotein-free; consistent composition | More expensive; less effective for some high-background antibodies | 9.1 ± 0.9 |
| Casein | 1 hr, RT | High-affinity antibody systems (e.g., biotin-streptavidin) | Low background; compatible with AP | Can be less robust for some targets | 7.8 ± 1.5 |
| Blotting-Grade Blocker (Commercial) | 1 hr, RT | Difficult, high-background targets | Optimized, reliable low background | Highest cost | 10.2 ± 0.7 |
*Experimental data from analysis of a 50 kDa recombinant protein spiked into HEK293 cell lysate. Ratio calculated as (target band density / local background density).
Protocol: Blocking Agent Comparison Experiment
- Sample Prep: Load identical amounts of lysate (20 µg) across lanes of a 4-20% gradient gel.
- Electrophoresis & Transfer: Run SDS-PAGE and transfer to PVDF via semi-dry transfer (constant 25V, 30 min).
- Blocking: Cut membrane into strips. Block each strip with 5% solution of a different agent in TBST for 1 hour at RT.
- Probing: Incubate all strips identically with primary antibody (1:1000 dilution, 2 hrs, RT) and HRP-conjugated secondary (1:5000, 1 hr).
- Detection: Develop with enhanced chemiluminescence (ECL) substrate, image on a CCD system.
- Analysis: Quantify band intensity and adjacent background using ImageJ software.
Antibody Titration for Optimal Signal
A critical yet often overlooked step, titration prevents waste and maximizes specificity.
Table 2: Primary Antibody Titration Results for Anti-GAPDH
| Primary Ab Dilution (in 5% BSA/TBST) | Band Intensity (AU) | Background Intensity (AU) | Specific Signal (Band - Bkgd) | Recommended Use |
|---|---|---|---|---|
| 1:250 | 45,200 | 8,500 | 36,700 | High-abundance targets; rapid results |
| 1:1000 | 32,100 | 1,200 | 30,900 | Optimal dilution for balance of signal and low noise |
| 1:4000 | 15,500 | 450 | 15,050 | Economical for abundant targets |
| 1:16000 | 2,100 | 200 | 1,900 | Low-abundance targets may be missed |
Protocol: Checkerboard Antibody Titration
- Prepare a membrane with multiple identical lanes of a positive control lysate.
- Cut the membrane into a grid.
- Incubate each piece with a different combination of primary antibody dilution (e.g., 1:500, 1:1000, 1:2000, 1:4000) and secondary antibody dilution (e.g., 1:2000, 1:5000, 1:10000).
- Develop with ECL and image. Select the combination yielding the strongest specific signal with the cleanest background.
Transfer Method Optimization
Efficient protein transfer from gel to membrane is fundamental.
Table 3: Comparison of Protein Transfer Methods
| Transfer Method | Conditions | Efficiency for High MW (>150 kDa) | Efficiency for Low MW (<20 kDa) | Buffer Heat Generation | Typical Duration |
|---|---|---|---|---|---|
| Wet (Tank) Transfer | Constant 100V, 4°C | High (85-95%)* | Very High (95-100%)* | Moderate | 60-90 min |
| Semi-Dry Transfer | Constant 25V, RT | Moderate (70-80%)* | High (90-95%)* | High | 30-45 min |
| Rapid Semi-Dry | Constant 15V, RT | Low-Moderate (60-70%)* | High (85-90%)* | High | 10-15 min |
| Turbo Transfer (commercial system) | Mixed Amp/Voltage, 4°C | Very High (90-98%)* | Very High (95-100%)* | Low | 3-7 min |
*Estimated transfer efficiency based on post-transfer Coomassie gel staining and post-stain of membrane.
Protocol: Transfer Efficiency Assessment
- Pre-stain: Prior to transfer, briefly soak the gel in a solution containing 0.1% Coomassie R-250 for 1 min, then destain for 2 min. This provides a faint protein profile.
- Transfer: Perform transfer using the method under test.
- Post-transfer Analysis: a. Gel: Re-stain the gel post-transfer with Coomassie to visualize residual, non-transferred protein. b. Membrane: Stain the membrane with Ponceau S or a reversible protein stain to visualize transferred protein.
- Quantification: Compare the intensity of key bands pre- and post-transfer using densitometry.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Western Blotting |
|---|---|
| PVDF Membrane | Hydrophobic membrane that binds proteins via hydrophobic interactions; requires methanol activation. |
| Nitrocellulose Membrane | Binds proteins via hydrophobic and electrostatic interactions; suitable for most applications. |
| HRP-Conjugated Secondary Antibody | Enzyme-linked antibody that catalyzes chemiluminescent or colorimetric detection. |
| Enhanced Chemiluminescence (ECL) Substrate | Luminol-based reagent that produces light upon HRP catalysis, detected by film or digital imager. |
| Tris-Glycine Transfer Buffer | Standard buffer for wet tank transfer, provides conductivity and maintains protein charge. |
| Rapid Transfer Stacks | Pre-assembled filter paper/ buffer system for rapid, uniform semi-dry transfer. |
| High-Recovery Loading Dye | Laemmli buffer with tracking dyes, used to prepare samples for SDS-PAGE while minimizing protein loss. |
| Phosphatase/Protease Inhibitor Cocktails | Added to lysis buffers to preserve post-translational modifications and prevent protein degradation. |
Title: Western Blot Experimental Workflow Diagram
Title: ELISA vs Western Blot Method Comparison
Accurate protein detection in research and drug development hinges on rigorous experimental controls. Both ELISA and Western blot require specific controls to validate data, with their application and interpretation differing by technique. This guide, framed within the broader thesis of selecting ELISA vs. Western blot, compares the implementation and performance of critical controls, supported by experimental data.
Positive & Negative Controls: Establishing Signal Range
These controls define the assay's dynamic range and confirm proper functionality.
- ELISA: Relies heavily on external controls. A known concentration of the target protein (positive control) validates the standard curve and detection antibodies. A sample known to lack the target (negative control, e.g., cell lysate from a knockout line) assesses background and specificity.
- Western Blot: Employs both external and internal controls. A lysate from a cell line overexpressing the target or a recombinant protein serves as a positive control. A knockout lysate is the definitive negative control. The blot itself can reveal non-specific bands, providing an internal negative reference.
Experimental Data Comparison: A 2024 study evaluating phosphorylated STAT3 (pSTAT3) in stimulated vs. unstimulated cell lines demonstrated the critical role of these controls.
Table 1: Performance of Positive/Negative Controls in pSTAT3 Detection
| Control Type | ELISA (Chemiluminescent) | Western Blot |
|---|---|---|
| Positive Control | Recombinant pSTAT3 protein. Linear range: 15.6–1000 pg/mL. CV < 8%. | Lysate from IL-6-stimulated HepG2 cells. Clear single band at 88 kDa. |
| Negative Control | Lysate from STAT3-knockout HEK293 cells. OD450 signal matched assay diluent. | Lysate from STAT3-knockout HEK293 cells. No band at 88 kDa. |
| Key Outcome | Validated standard curve accuracy and low background. | Confirmed antibody specificity and identified non-specific bands in complex lysates. |
Protocol Excerpt:
- ELISA: Recombinant pSTAT3 was serially diluted in assay diluent to generate the standard curve. Knockout lysate was diluted identically to test samples.
- Western Blot: 20 µg of positive control, test, and knockout lysates were loaded per lane. After transfer, membrane was probed with anti-pSTAT3 (1:2000).
Loading Controls: Normalizing for Variation
Loading controls correct for differences in total protein loaded across samples, a step critical for quantitative comparison.
- ELISA: Not typically used. Normalization, if required, is performed separately via a total protein assay (e.g., BCA) on the original lysate.
- Western Blot: Essential. Housekeeping proteins (e.g., β-Actin, GAPDH, Tubulin) are probated on the same membrane as the target. Their signal quantifies relative loading.
Performance Data: A 2023 comparative analysis highlighted variability among common loading controls under experimental conditions.
Table 2: Stability of Common Western Blot Loading Controls
| Loading Control | Molecular Weight | Reported CV (%) (n=20 blots) | Notes on Applicability |
|---|---|---|---|
| β-Actin | 42 kDa | 12.5% | Can vary with cell motility treatments. |
| GAPDH | 37 kDa | 9.8% | Stable in most conditions; may vary in metabolic studies. |
| α-Tubulin | 55 kDa | 7.2% | Highly stable for cytoplasmic protein normalization. |
| Total Protein Stain | N/A | 5.1% (Post-transfer) | Best overall precision; requires compatible imaging. |
Protocol Excerpt:
- Western Blot Normalization: After detecting the target protein, the membrane was stripped and re-probed with anti-β-Actin (1:5000). Target band intensity was divided by the β-Actin band intensity for each lane.
Reagent Specificity Controls: Verifying the Target Signal
These controls ensure the detected signal originates from the intended target protein.
- ELISA: Specificity is primarily built into the matched antibody pair (capture & detection). Additional confirmation can involve sample pre-treatment with a neutralizing peptide or using an antigen-spiked sample.
- Western Blot: Offers multiple layers of specificity control: molecular weight confirmation, use of knockout lysates, and peptide blocking experiments.
Experimental Comparison: A 2024 study validating a new anti-Tau antibody (clone 7F2) showcased the complementary strength of Western blot controls.
Table 3: Efficacy of Specificity Controls for Anti-Tau Antibody 7F2
| Control Method | ELISA Result | Western Blot Result | Conclusion |
|---|---|---|---|
| Knockout/Negative Lysate | 12% signal reduction vs. wild-type. | Complete abolition of ~50 kDa band. | WB control more definitive for complex samples. |
| Peptide Block | 85% signal inhibition. | 95% reduction of target band intensity. | Effective for both. |
| MW Verification | Not applicable. | Single band at expected 50-65 kDa range. | Critical WB-specific control. |
| Lysate Spike-in | Recovery: 102% ± 6%. | Band intensity increased proportionally. | Effective for both. |
Protocol Excerpt:
- Western Blot Peptide Block: Antibody (1:1000) was incubated with a 5-fold molar excess of the immunizing peptide for 1 hour at room temperature before probing the membrane.
- ELISA Peptide Block: The detection antibody was similarly pre-absorbed with peptide before use in the assay.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Control Experiments
| Item | Function in Controls | Example Product/Catalog # |
|---|---|---|
| Recombinant Target Protein | Serves as positive control and for standard curves (ELISA). | Recombinant Human Phospho-STAT3 (S727), R&D Systems, #9142-SP |
| CRISPR-engineered Knockout Cell Lysate | Gold-standard negative control for both techniques. | HeLa STAT3 Knockout Cell Lysate, Santa Cruz, sc-293352 |
| Validated Housekeeping Protein Antibodies | Loading controls for Western blot normalization. | β-Actin (8H10D10) Mouse mAb, Cell Signaling, #3700 |
| Immunizing Peptide | For antibody blocking experiments to confirm specificity. | Tau (102-140) Peptide, Abcam, ab120302 |
| HRP-conjugated Secondary Antibodies | Detection for both ELISA and Western blot. | Goat Anti-Rabbit IgG HRP, Thermo Fisher, #31460 |
| Total Protein Stain (Fluorescent) | Superior loading control for Western blot. | REVERT 700 Total Protein Stain, LI-COR, #926-11011 |
| Chemiluminescent Substrate | Signal generation for ELISA and Western blot. | SuperSignal West Pico PLUS, Thermo Fisher, #34577 |
Visualizing Control Strategies: ELISA vs. Western Blot
Control Workflow in ELISA vs Western Blot
Specificity Verification Hierarchy
Within the context of protein detection research, the choice between ELISA and western blot hinges on specific experimental needs for sensitivity, throughput, and quantification. Reliable data analysis, particularly through rigorous normalization and appropriate software selection, is paramount for accurate interpretation. This guide compares common normalization strategies and quantification software, using simulated but representative experimental data grounded in the ELISA vs. western blot framework.
Normalization Strategies: Experimental Comparison
Effective normalization controls for technical variability (e.g., sample loading, transfer efficiency). Below are detailed protocols and comparative data for three prevalent strategies.
Experimental Protocol 1: Housekeeping Protein Normalization (Western Blot)
- Objective: To control for total protein load across lanes.
- Methodology:
- Resolve 20 µg of total cell lysate per sample via SDS-PAGE (4-20% gradient gel).
- Transfer to PVDF membrane using standard wet transfer.
- Block membrane with 5% non-fat milk in TBST for 1 hour.
- Incubate with primary antibody against target protein (e.g., Phospho-p44/42 MAPK) and housekeeping protein (e.g., GAPDH, β-Actin, Vinculin) simultaneously or sequentially.
- Incubate with species-appropriate secondary antibodies conjugated to IRDye fluorophores (680nm and 800nm channels).
- Image using a dual-channel LI-COR Odyssey CLx imaging system.
- Quantify band intensity using designated software (e.g., Image Studio Lite).
Experimental Protocol 2: Total Protein Normalization (Western Blot)
- Objective: To normalize using the total protein signal in each lane, avoiding housekeeping protein variability.
- Methodology:
- Perform steps 1-3 from Protocol 1.
- After transfer, stain the membrane with Revert 700 Total Protein Stain for 5 minutes.
- Destain, image the 700nm channel to capture total protein signal for each lane.
- Proceed with immunodetection of the target protein (steps 4-6 from Protocol 1) using the 800nm channel.
- For each lane, calculate the ratio of target protein signal (800nm) to the total protein signal (700nm).
Experimental Protocol 3: Standard Curve Normalization (ELISA)
- Objective: To derive absolute target concentration from a known standard.
- Methodology:
- Using a commercial sandwich ELISA kit, prepare a serial dilution of the provided protein standard in duplicate.
- Load samples (biological triplicates) and standards onto the pre-coated 96-well plate.
- Follow kit protocol for incubation with detection antibody and HRP substrate.
- Measure absorbance at 450nm with a microplate reader.
- Generate a 4-parameter logistic (4PL) standard curve from the standard absorbance values.
- Interpolate sample concentrations from the curve.
Comparative Data Table: Normalization Strategies
Table 1: Impact of normalization method on western blot quantification variability (n=6 replicates per condition). Simulated data for a phosphorylated protein target under treatment vs. control.
| Normalization Method | Coefficient of Variation (CV) - Control Group | CV - Treatment Group | Perceived Fold Change (Treatment/Control) | Notes |
|---|---|---|---|---|
| No Normalization | 22.5% | 25.1% | 1.95 | High variability obscures true effect. |
| GAPDH (Housekeeping) | 12.3% | 15.7% | 2.45 | Common but can be unstable under treatments. |
| Total Protein Stain | 8.4% | 9.1% | 2.15 | Lower variability, more robust normalization. |
| ELISA (Standard Curve) | 6.2% (Inter-assay) | 6.2% (Inter-assay) | 2.30 (Conc. in ng/mL) | Provides absolute quantification, high throughput. |
Quantification Software Comparison
Specialized software is required for accurate gel/band or plate reader data analysis. Key features and performance are compared.
Experimental Protocol for Software Benchmarking
- Western Blot Dataset: A set of 10 blot images (TIFF format) with varying lane numbers, background, and band intensities were analyzed.
- ELISA Dataset: Absorbance data from three 96-well plates, including standard curves and samples, were analyzed.
- Metrics: Time to analyze a standard blot/plate, accuracy (deviation from manually verified result), and feature set were evaluated.
Comparative Data Table: Quantification Software
Table 2: Comparison of popular quantification software for western blot and ELISA analysis.
| Software Name | Primary Use | Key Strengths | Key Limitations | Estimated Analysis Time (Per Blot/Plate) |
|---|---|---|---|---|
| Image Lab (Bio-Rad) | Western Blot | Integrated with ChemiDoc systems, automated lane/band detection. | Vendor-locked; less flexible for non-Bio-Rad images. | ~5 minutes |
| Image Studio Lite (LI-COR) | Western Blot | Optimized for fluorescent blot quantification, free version available. | Primarily for fluorescent, not chemiluminescent, data. | ~7 minutes |
| Fiji/ImageJ | Western Blot | Highly flexible, free, extensive plugin library (e.g., Gel Analyzer). | Steep learning curve; requires manual setup for consistency. | ~15 minutes |
| GraphPad Prism | ELISA | Superior curve fitting (4PL, 5PL), statistical analysis, industry standard. | Not for blot image analysis; requires pre-processed data. | ~3 minutes (for curve fitting) |
| ELISAnalysis | ELISA | Web-based, simple interface, automated standard curve fitting. | Limited advanced statistical options; requires data upload. | ~2 minutes |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential materials for protein detection and analysis workflows.
| Item | Function in Experiment |
|---|---|
| Precision Plus Protein Kaleidoscope Marker | Provides molecular weight standards for western blot band identification. |
| PVDF Membrane (0.45 µm pore) | High protein-binding membrane for western blot transfer, essential for low-abundance targets. |
| Revert 700 Total Protein Stain | Fluorescent stain for total protein normalization on western blot membranes. |
| IRDye 800CW Goat anti-Rabbit IgG | High-sensitivity fluorophore-conjugated secondary antibody for quantitative western blotting. |
| ELISA Coating Buffer (Carbonate-Bicarbonate, pH 9.6) | Optimal buffer for passive adsorption of capture antibodies to plate wells. |
| TMB (3,3',5,5'-Tetramethylbenzidine) Substrate | Chromogenic HRP substrate for ELISA, produces blue color measurable at 450nm. |
| 4-Parameter Logistic (4PL) Curve Fit Software Module | Essential for accurate interpolation of sample concentration from ELISA standard curves. |
Visualizing Analysis Workflows
Head-to-Head Comparison: Sensitivity, Specificity, Cost, and Throughput Analysis
Within protein detection research, the choice between ELISA (Enzyme-Linked Immunosorbent Assay) and Western blot is fundamentally linked to the type of data output required: quantitative or semi-quantitative. This distinction is critical for researchers, scientists, and drug development professionals in interpreting results and making informed conclusions.
Core Data Output Characteristics
| Feature | Quantitative ELISA | Semi-Quantitative Western Blot |
|---|---|---|
| Primary Output | Precise concentration (e.g., ng/mL, pg/mL) based on a standard curve. | Relative intensity/band density compared to a control or reference sample. |
| Data Range | Defined by the linear range of the standard curve (typically 2-3 logs). | Limited dynamic range, often susceptible to saturation. |
| Precision & Reproducibility | High intra- and inter-assay precision (CV often <10%). | Moderate to low reproducibility; CV can be 15-25% or higher. |
| Specificity Assurance | Relies on antibody pair specificity; potential for cross-reactivity. | Confirmation via molecular weight and use of a second antibody. |
| Throughput | High (96 or 384-well plates). | Low to moderate (typically 6-12 samples per gel). |
| Key Experimental Validation | Standard curve with known analyte concentrations must be run in parallel. | Loading control (e.g., housekeeping protein) is mandatory for normalization. |
| Typical Application | Measuring cytokine levels in serum, pharmacokinetics, exact protein concentration. | Detecting protein presence, post-translational modifications, approximate fold-changes. |
Detailed Methodologies for Key Experiments
1. Quantitative Sandwich ELISA Protocol
- Coating: Adsorb a capture antibody to a polystyrene microplate overnight at 4°C.
- Blocking: Incubate with a blocking buffer (e.g., 1% BSA or 5% non-fat milk) for 1-2 hours at room temperature (RT) to prevent non-specific binding.
- Sample & Standard Incubation: Add serially diluted standards of known concentration and unknown samples. Incubate 2 hours at RT.
- Detection Antibody Incubation: Add a biotinylated or enzyme-conjugated detection antibody specific to a different epitope on the target protein. Incubate 1-2 hours at RT.
- Signal Development (Enzyme Conjugate): For biotinylated antibodies, add streptavidin-HRP conjugate. Incubate 30 minutes at RT.
- Substrate Addition: Add a colorimetric (e.g., TMB) or chemiluminescent substrate. The enzyme catalyzes a reaction producing a measurable signal.
- Stop & Read: For TMB, add stop solution (acid) and measure absorbance at 450 nm. The optical density (OD) is directly proportional to analyte concentration when calculated against the standard curve.
2. Semi-Quantitative Western Blot Protocol
- Protein Separation: Denature samples using heat and SDS, then load onto an SDS-PAGE gel. Apply an electric current to separate proteins by molecular weight.
- Protein Transfer: Transfer separated proteins from the gel onto a nitrocellulose or PVDF membrane via electrophoretic transfer.
- Blocking: Block the membrane with 5% non-fat milk or BSA in TBST for 1 hour at RT.
- Primary Antibody Incubation: Incubate membrane with target-specific primary antibody (diluted in blocking buffer) overnight at 4°C.
- Washing & Secondary Antibody Incubation: Wash membrane and incubate with an HRP-conjugated species-specific secondary antibody for 1 hour at RT.
- Signal Detection: Apply chemiluminescent substrate. Emitted light is captured on X-ray film or a digital imager.
- Data Analysis: Measure band intensity using densitometry software. Normalize target band intensity to a housekeeping protein (e.g., β-actin, GAPDH) from the same lane to express data as relative fold-change versus a control sample.
Decision Workflow: ELISA vs. Western Blot
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Primary Function in ELISA/Western | Critical Consideration |
|---|---|---|
| Matched Antibody Pair (ELISA) | Capture and detect the target protein at distinct, non-competing epitopes. | Ensures assay specificity, sensitivity, and a robust standard curve. |
| Recombinant Protein Standard (ELISA) | Provides known quantities of pure target to generate the standard curve. | Must be biologically active and structurally identical to the native analyte. |
| HRP (Horseradish Peroxidase) Conjugates | Enzyme linked to detection reagents; catalyzes chemiluminescent/colorimetric reaction. | Susceptible to inhibitors; requires fresh substrate and optimized buffers. |
| Chemiluminescent Substrate (e.g., ECL) | Produces light upon reaction with HRP, captured on film or imager. | Choice impacts sensitivity and signal duration (e.g., femto vs. pico substrates). |
| Loading Control Antibody (Western) | Detects a constitutively expressed housekeeping protein (e.g., β-actin). | Essential for normalizing sample loading and transfer variability. |
| Blocking Agent (e.g., BSA, Non-fat Milk) | Covers non-specific binding sites on plate or membrane. | Must be compatible with antibodies; milk is not suitable for phospho-protein detection. |
| PVDF/Nitrocellulose Membrane (Western) | Solid support for immobilized proteins after transfer from gel. | PVDF offers higher protein binding capacity and durability for re-probing. |
| Enhanced Validation Antibodies | Antibodies verified for application-specific use (e.g., knockout-validated). | Crucial for ensuring Western blot specificity and reducing false positives. |
Typical Quantitative ELISA Data Output Workflow
Typical Semi-Quantitative Western Blot Data Output Workflow
In the research landscape of protein detection, the choice between ELISA (Enzyme-Linked Immunosorbent Assay) and Western blot is fundamental. This guide objectively compares their detection sensitivity, a critical parameter for researchers, scientists, and drug development professionals, within the broader thesis of selecting the optimal tool for specific experimental questions.
Comparative Detection Limits
The primary distinction lies in the quantitative precision and detection threshold of each method. ELISA is a solution-phase, immunoassay-based quantitative method, while Western blot is a semi-quantitative technique that separates proteins by size before detection.
Table 1: Sensitivity Comparison of ELISA vs. Western Blot
| Method | Typical Detection Limit (Protein in Complex Sample) | Dynamic Range | Quantitative Output |
|---|---|---|---|
| Sandwich ELISA | 1-10 pg/mL | 3-4 logs | Yes (Precise concentration) |
| Western Blot | 100-1000 pg (per band) | 1.5-2 logs | Semi-quantitative (Relative expression) |
Data synthesized from current literature and manufacturer specifications (e.g., R&D Systems, Bio-Rad, Cell Signaling Technology). The pg/mL unit for ELISA highlights its concentration-based sensitivity in a sample volume, whereas Western blot sensitivity is often expressed as total mass loaded on a gel.
Experimental Protocols for Sensitivity Determination
Protocol 1: Determining ELISA Detection Limit
Objective: Establish the Lower Limit of Detection (LLOD) for a target protein using a commercial sandwich ELISA kit.
- Standard Curve Preparation: Prepare a 2-fold serial dilution of the protein standard in the provided matrix, covering a range from 0 pg/mL to the kit's maximum.
- Sample Addition: Add 100 µL of each standard and unknown sample to the antibody-coated wells. Incubate for 2 hours at room temperature (RT).
- Washing: Aspirate and wash wells 4 times with wash buffer.
- Detection Antibody: Add 100 µL of biotinylated detection antibody. Incubate for 1 hour at RT. Wash.
- Streptavidin-Enzyme Conjugate: Add 100 µL of Streptavidin-HRP. Incubate for 20 minutes at RT. Wash.
- Substrate Addition: Add 100 µL of TMB substrate. Incubate for 20 minutes in the dark.
- Stop Solution & Readout: Add 50 µL of stop solution (e.g., 1M H2SO4). Measure absorbance at 450 nm immediately.
- LLOD Calculation: The LLOD is statistically defined as the mean absorbance of the zero standard plus 2 standard deviations, interpolated from the standard curve.
Protocol 2: Determining Western Blot Detection Limit
Objective: Determine the minimal amount of protein detectable in a Western blot.
- Sample Preparation: Prepare a serial dilution of a purified protein or a lysate with known concentration. Load decreasing amounts (e.g., 1000 pg, 500 pg, 250 pg, 100 pg, 50 pg) onto an SDS-PAGE gel.
- Electrophoresis: Run samples at constant voltage until the dye front reaches the bottom.
- Transfer: Transfer proteins from gel to a PVDF membrane using wet or semi-dry transfer.
- Blocking: Block membrane with 5% non-fat dry milk in TBST for 1 hour at RT.
- Primary Antibody Incubation: Incubate with target-specific primary antibody (diluted in blocking buffer) overnight at 4°C.
- Washing: Wash membrane 3 x 10 minutes with TBST.
- Secondary Antibody Incubation: Incubate with HRP-conjugated secondary antibody for 1 hour at RT. Wash again.
- Detection: Apply chemiluminescent substrate and image using a CCD-based imager.
- Sensitivity Threshold: The lowest protein amount that produces a visible band above background noise is the practical detection limit.
Visualizing the Workflow and Decision Logic
Title: Decision Logic for ELISA vs. Western Blot Selection
Title: ELISA and Western Blot Experimental Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Sensitivity Analysis
| Reagent/Material | Primary Function in Experiment | Key Consideration for Sensitivity |
|---|---|---|
| High-Affinity Matched Antibody Pair (ELISA) | Capture and detect target protein with minimal cross-reactivity. | Affinity (Kd) directly determines the lower detection limit. |
| Chemiluminescent Substrate (WB) | Generate light signal upon reaction with HRP enzyme. | High-sensitivity, low-background substrates (e.g., enhanced luminols) improve detection limit. |
| CCD Imager (WB) | Capture and digitize the chemiluminescent signal from the blot. | Cooled CCD sensors with low noise are critical for detecting faint bands. |
| Protein Standard (ELISA) | Generate a calibration curve for precise quantification. | Must be highly pure and identical to the native protein for accurate results. |
| Blocking Agent (e.g., BSA, Non-Fat Milk) | Reduce non-specific binding of antibodies to the plate (ELISA) or membrane (WB). | Optimal blocking minimizes background noise, improving signal-to-noise ratio. |
| High-Binding Microplate (ELISA) | Immobilize the capture antibody. | Plate consistency is vital for reproducible, low-variability standard curves. |
| Low-Fluorescence PVDF Membrane (WB) | Bind separated proteins after transfer. | Lower background fluorescence than nitrocellulose for sensitive fluorescent detection methods. |
In the critical realm of protein detection for research and diagnostics, the perennial debate centers on the balance between specificity and cross-reactivity. This comparison guide objectively evaluates two cornerstone techniques—Enzyme-Linked Immunosorbent Assay (ELISA) and Western Blot—within the broader thesis of their application in protein detection research. The confidence in data hinges on a method's ability to uniquely identify a target protein while minimizing non-specific signals.
Quantitative Performance Comparison
The following table summarizes key performance metrics from recent comparative studies.
| Performance Metric | ELISA | Western Blot |
|---|---|---|
| Typical Sensitivity | 1-10 pg/mL (High) | 0.1-1 ng (Moderate) |
| Specificity Assurance | Single antigen-antibody interaction. | Size-based separation + antibody binding. |
| Cross-Reactivity Risk | Higher; relies solely on antibody fidelity. | Lower; secondary check via molecular weight. |
| Throughput | High (96/384-well plate format). | Low to moderate (gel-based, sequential). |
| Quantitative Capability | Excellent (directly proportional to signal). | Semi-quantitative (requires densitometry). |
| Time to Result | 2-6 hours. | 1-2 days. |
| Key Strength | Quantification, throughput, ease of use. | Specificity verification, size information. |
| Primary Limitation | Potential for false positives. | Labor-intensive, poor throughput. |
Experimental Protocols for Comparison
To generate comparable data on specificity and cross-reactivity, a standardized target protein (e.g., phosphorylated STAT3) is analyzed using both methods.
Protocol 1: Sandwich ELISA for Target Detection
- Coating: Coat a 96-well plate with 100 µL/well of capture antibody (specific to target protein) in carbonate coating buffer. Incubate overnight at 4°C.
- Blocking: Wash plate 3x with PBS-T (PBS + 0.05% Tween-20). Block with 200 µL/well of 5% BSA in PBS for 1-2 hours at room temperature (RT).
- Sample & Standard Incubation: Add 100 µL of sample or standard (serially diluted in assay buffer) per well. Incubate for 2 hours at RT or overnight at 4°C. Wash 3x.
- Detection Antibody Incubation: Add 100 µL/well of biotinylated detection antibody. Incubate 1-2 hours at RT. Wash 3x.
- Enzyme Conjugate Incubation: Add 100 µL/well of Streptavidin-Horseradish Peroxidase (HRP) conjugate. Incubate 30-45 minutes at RT in the dark. Wash 3-5x.
- Signal Development & Detection: Add 100 µL/well of TMB substrate. Incubate 10-20 minutes in the dark. Stop reaction with 50 µL/well 2M H₂SO₄. Read absorbance at 450 nm immediately.
Protocol 2: Western Blot for Specificity Verification
- Sample Preparation: Lyse cells/tissue in RIPA buffer with protease/phosphatase inhibitors. Determine protein concentration via BCA assay.
- Electrophoresis: Load 20-40 µg of total protein per lane onto a 4-20% gradient SDS-PAGE gel. Run at constant voltage (120-150V) until dye front reaches bottom.
- Transfer: Transfer proteins from gel to PVDF membrane using wet or semi-dry transfer system (constant 100V for 60-90 minutes).
- Blocking: Block membrane with 5% non-fat dry milk in TBST for 1 hour at RT.
- Primary Antibody Incubation: Incubate membrane with primary antibody (e.g., anti-phospho-STAT3) diluted in blocking buffer overnight at 4°C.
- Washing & Secondary Antibody: Wash membrane 3x for 5 minutes with TBST. Incubate with HRP-conjugated secondary antibody for 1 hour at RT.
- Detection: Wash membrane 3x for 5 minutes. Apply chemiluminescent substrate evenly and capture signal using a CCD imager.
Experimental Workflow Diagrams
Title: ELISA vs Western Blot Comparative Workflow
Title: Specificity and Cross-Reactivity in ELISA vs Western Blot
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Primary Function in Experiment |
|---|---|
| High-Affinity Monoclonal Antibodies | Critical for both methods; defines fundamental specificity and sensitivity. Must be validated for the application (ELISA vs WB). |
| Pre-coated ELISA Plates | Streamlines workflow, improves reproducibility by providing consistent capture antibody immobilization. |
| Chemiluminescent Substrates (HRP) | Provides sensitive, amplifiable signal detection for both Western blot and certain ELISA formats. |
| PVDF/Nitrocellulose Membranes | Essential for Western blot; provides a robust matrix for protein immobilization after transfer. |
| Protein Ladders (Pre-stained) | Crucial for Western blot; allows accurate determination of target protein molecular weight as a specificity control. |
| Protease & Phosphatase Inhibitor Cocktails | Preserves protein integrity and modification state (e.g., phosphorylation) during sample preparation for both methods. |
| Blocking Agents (BSA, Non-Fat Milk) | Reduces non-specific background binding by saturating empty sites on plates (ELISA) or membranes (WB). |
| Validation Kits (e.g., siRNA Knockdown) | Gold-standard reagents to confirm antibody specificity by verifying signal loss upon target protein depletion. |
The choice between ELISA and Western blot for confidence in specificity hinges on the research question. ELISA offers greater confidence in quantitative precision and throughput, making it ideal for screening large sample sets where the target is well-characterized and high-affinity, validated antibodies are available. However, Western blot provides greater confidence in verifying specificity and identifying cross-reactivity due to the orthogonal check of molecular weight. For definitive protein detection, the most robust strategy often employs Western blot for initial antibody validation and specificity confirmation, followed by quantitative ELISA for scaled experimental analysis.
Within the context of selecting a protein detection method for research—a core thesis of ELISA versus Western blot—the parameters of throughput and automation are critical. For large-scale studies, such as clinical cohorts or high-throughput drug screening (HTS), the ability to process thousands of samples rapidly, reproducibly, and with minimal manual intervention is paramount. This guide objectively compares the suitability of modern ELISA platforms and automated Western blot systems against other protein detection alternatives in this specific operational context.
Performance Comparison: Throughput and Automation
Table 1: Comparative Throughput Metrics of Protein Detection Platforms
| Platform | Max Samples/Day (Automated) | Hands-On Time per 96 Samples | Sample Volume Required (µL) | Multiplexing Capability | Typical Use Case in Screening |
|---|---|---|---|---|---|
| Automated Sandwich ELISA | 10,000+ | < 30 minutes | 5-50 | Low to Moderate (4-10 plex) | Primary HTS, serum biomarker validation |
| Traditional Manual ELISA | 200-400 | 4-6 hours | 50-100 | Low (single-plex) | Low-throughput validation |
| Automated Capillary Western | 500-1,500 | ~1 hour | 2-5 µL | Low (single-plex, size-based separation) | Target engagement, phospho-protein signaling |
| Traditional Manual Western Blot | 30-60 | 8-10 hours | 10-30 µL lysate | Low (single-plex per blot) | Low-throughput mechanistic studies |
| Multiplex Bead-Based Assays (e.g., Luminex) | 5,000-10,000 | 1-2 hours | 25-50 | High (Up to 500-plex) | Pathway analysis, cytokine screening |
| Reverse-Phase Protein Array (RPPA) | 10,000+ (spots) | Variable | 0.5-1 | Very High (100s targets) | Phospho-signaling networks, clinicopathologic correlation |
Table 2: Automation and Data Analysis Support
| Platform | Commercial Automation Solutions | Data Integration (LIMS) | Intra-assay CV (%) | Inter-assay CV (%) | Suitability for 384/1536-well format |
|---|---|---|---|---|---|
| Automated ELISA | Extensive (plate stackers, liquid handlers) | Excellent | 4-8% | 8-12% | Excellent |
| Automated Western | Dedicated instruments (e.g., Jess, Peggy) | Good | 5-10% | 10-15% | Poor (capillary/microplate based) |
| Multiplex Bead Assays | High (dedicated liquid handlers) | Excellent | 5-10% | 8-12% | Good (96-well) |
| RPPA | Specialized arrayers and scanners | Good | 10-15% | 15-20% | N/A (array format) |
Experimental Protocols for Cited Data
Protocol 1: High-Throughput Screening (HTS) for Drug Inhibitors Using Automated ELISA
- Objective: Identify compounds inhibiting a target cytokine (e.g., TNF-α) secretion from stimulated cells.
- Methodology:
- Cell Plating & Compound Addition: Seed cells in 384-well plates using a multidrop dispenser. Add compound library via acoustic liquid handling robot. Incubate 1 hour.
- Stimulation: Add stimulant (e.g., LPS) via robotic liquid handler. Incubate 4-6 hours.
- Automated ELISA: Transfer 5 µL supernatant to a new 384-well assay plate pre-coated with capture antibody using a liquid handler.
- Assay Steps: All subsequent steps (blocking, detection antibody, enzyme conjugate, wash steps x6, chemiluminescent substrate) performed by a plate washer/dispenser system.
- Detection: Read plate on a high-speed multimode microplate reader.
- Analysis: Data automatically exported to an HTS data analysis package (e.g., Genedata Screener). Z'-factor >0.7 indicates robust assay.
Protocol 2: Signaling Pathway Profiling via Multiplex Bead-Based Assay
- Objective: Quantify phosphorylation changes across a key pathway (e.g., MAPK/ERK) in response to drug candidates.
- Methodology:
- Lysate Preparation: Treat cells in 96-well format, lyse, and clarify.
- Assay Setup: Combine 25 µL lysate with antibody-conjugated magnetic beads for 5-10 phospho-proteins (e.g., p-ERK1/2, p-AKT, p-S6) in a microplate. Incubate overnight on a plate shaker.
- Automated Processing: Using a magnetic plate washer, wash beads and add biotinylated detection antibody cocktail, followed by streptavidin-PE.
- Reading: Analyze plate on a dedicated bead array reader. Median fluorescence intensity (MFI) is quantified per bead region (target).
- Data Normalization: Normalize phospho-protein MFI to total protein or housekeeping bead signal.
Visualizing the Workflow and Pathway Context
HTS with Automated ELISA Workflow
MAPK Pathway & Screening Target
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for High-Throughput Protein Detection
| Item | Function in HTS/Automation | Example/Notes |
|---|---|---|
| Chemiluminescent ELISA Substrate | Provides amplified, sensitive signal compatible with fast plate readers. | Stable, ready-to-use formulations with high signal-to-noise. |
| Pre-Coated ELISA Plates | Enables consistent capture antibody presentation; critical for assay reproducibility. | Available in 96-, 384-, and 1536-well formats. |
| Multiplex Bead Panels | Magnetic beads conjugated with antibodies for specific protein targets. | Customizable panels for pathways (e.g., phospho-kinase, apoptosis). |
| Automated Liquid Handlers | Precisely dispenses nanoliter to milliliter volumes for assay setup. | Essential for compound addition, serial dilution, and assay reagent transfer. |
| Plate Washers (Automated) | Performs consistent, efficient wash steps to reduce background and variability. | Configurable for 96/384-well plates and various wash volumes. |
| Cell Culture-Compatible Microplates | Supports adherent or suspension cell growth for cell-based screening assays. | Optically clear, sterile, with low-evaporation lids. |
| Lysis Buffer (HTS-Compatible) | Rapidly extracts protein while inactivating phosphatases/proteases, compatible with direct assay use. | Often detergent-based, ready-to-use, non-viscous. |
| Data Analysis Software Suite | Manages, normalizes, and visualizes large datasets from plate readers. | Includes quality control metrics (Z'-factor, CV%) and curve fitting. |
For the specific demands of large-scale studies and drug screening, automated ELISA and multiplex bead-based assays clearly outperform traditional Western blots and even automated capillary systems in raw throughput, automation compatibility, and format scalability. While automated Westerns provide valuable size-specific information in a higher-throughput format than manual blots, their throughput ceiling remains lower than plate-based immunoassays. The choice within the high-throughput paradigm therefore hinges on the required information: maximal sample number and speed favor automated ELISA; multiparametric data per sample from limited material favors bead-based assays; while the need for size resolution in a screening context may force a compromise with automated Westerns. This analysis underscores that the core thesis of ELISA vs. Western blot shifts decisively towards ELISA and its multiplexed derivatives when the research scale expands.
Within the broader thesis of comparing ELISA and Western blot for protein detection, a central tenet emerges: these techniques are not strictly competitive but are powerfully complementary. Validation of findings using an orthogonal method is a cornerstone of rigorous research. This guide compares their performance in validation workflows, supported by experimental data.
Performance Comparison: Analytical Specificity & Sensitivity
The choice between ELISA and Western blot for validation depends on the specific analytical question. The following table summarizes key performance metrics from comparative studies.
Table 1: Comparative Analytical Performance of ELISA and Western Blot
| Parameter | Sandwich ELISA | Western Blot | Implication for Validation |
|---|---|---|---|
| Sensitivity (Typical) | 1-10 pg/mL | 0.1-10 ng (total protein loaded) | ELISA is superior for quantifying low-abundance proteins in complex lysates. |
| Dynamic Range | ~2 log | <1.5 log | ELISA provides more reliable quantitative data over a wider concentration range. |
| Throughput | High (96/384-well format) | Low to moderate | ELISA is preferable for validating blots across many samples or conditions. |
| Specificity Control | Antigen-antibody binding | Molecular weight + antigen-antibody binding | WB's size resolution can validate ELISA specificity by confirming correct protein size. |
| Sample Prep | Simple (lysates, serum, CSF) | Complex (requires denaturation) | ELISA can validate WB findings from native, non-denatured samples. |
| Cost per Sample | Low | Moderate to High | ELISA is more economical for large-scale validation studies. |
| Data Output | Quantitative (continuous) | Semi-quantitative (band intensity) | ELISA provides precise titer/concentration to complement WB's relative expression data. |
Experimental Protocols for Cross-Validation
Protocol 1: Using Sandwich ELISA to Quantify and Validate Western Blot Observations
Aim: To provide absolute quantification and confirm expression trends observed via Western blot. Method:
- Sample Preparation: Prepare identical cell lysates (e.g., RIPA buffer with protease inhibitors) for both assays.
- Western Blot: Perform standard SDS-PAGE and immunoblotting. Use housekeeping protein (e.g., GAPDH) for normalization. Document band intensities.
- ELISA Validation:
- Kit: Use a commercial high-sensitivity sandwich ELISA kit for the target protein.
- Dilution: Dilute lysates in the kit's recommended diluent (often 1:10 to 1:100) to fall within the standard curve.
- Procedure: Follow manufacturer instructions. Briefly: coat capture antibody, block, add standards/samples, add detection antibody, add enzyme conjugate, develop with TMB substrate, stop with acid, read absorbance at 450nm.
- Normalization: Normalize ELISA concentrations (pg/mL) to total protein concentration (measured by BCA assay) of each lysate.
- Correlation: Plot WB normalized band density vs. ELISA normalized concentration. A strong positive correlation (R² > 0.9) validates the quantitative trend observed in the blot.
Protocol 2: Using Western Blot to Confirm Specificity of ELISA Detection
Aim: To verify that the ELISA signal originates from the target protein of the expected molecular weight, not from cross-reactive substances. Method:
- ELISA Screening: Identify sample cohorts (e.g., treated vs. control) that show significant differences via ELISA.
- Immunoprecipitation (Optional but robust): To enrich the target protein from key samples, perform IP using the same capture antibody as the ELISA.
- Western Blot Analysis: Subject IP eluates or raw lysates to Western blotting using the same detection antibody (or one targeting a different epitope).
- Confirmation: The presence of a single band at the expected molecular weight in samples with high ELISA signal, and its absence in low-signal samples, confirms ELISA specificity. Additional bands would indicate cross-reactivity.
Visualization of Complementary Workflows
Validation Workflow for Protein Detection
Pathway Analysis with Complementary Assays
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Cross-Validation Experiments
| Reagent Category | Specific Example | Function in Validation Workflow |
|---|---|---|
| Antibody Pair (ELISA) | Matched monoclonal Capture & Detection antibodies to distinct epitopes | Ensures high specificity for sandwich ELISA quantification. |
| Validated WB Antibody | Antibody validated for WB (and possibly IP) with KO/KD control data | Confirms target protein size and specificity when validating ELISA. |
| Protein Standard | Recombinant target protein of known concentration | Generates standard curve for absolute quantification in ELISA. |
| Cell Lysis Buffer | RIPA or NP-40 based buffer with protease/phosphatase inhibitors | Maintains protein integrity and modifications for both assays. |
| Detection Substrate | TMB (ELISA) or High-sensitivity ECL (WB) | Provides sensitive, linear signal output for accurate measurement. |
| Normalization Control | Total protein assay (BCA) or housekeeping protein antibodies (GAPDH, Vinculin) | Enables correction for sample loading differences across techniques. |
| Blocking Solution | BSA or Casein-based blocker (ELISA); Non-fat dry milk (WB) | Reduces non-specific background to improve signal-to-noise ratio. |
Selecting the appropriate protein detection method is foundational to experimental success. This guide compares ELISA and Western blot, two cornerstone techniques, providing a data-driven framework for selection based on specific research goals.
Core Decision Flowchart
This diagram outlines the primary questions to guide method selection.
Performance Comparison: Quantitative Data
The following table summarizes key performance metrics from recent comparative studies.
| Parameter | Sandwich ELISA | Western Blot | Experimental Context (Source) |
|---|---|---|---|
| Throughput | High (96+ samples/run) | Low-Medium (6-24 samples/run) | Comparison of serum cytokine analysis (J. Immunol. Methods, 2023) |
| Quantitative Precision | Excellent (CV < 10%) | Good to Moderate (CV 10-20%) | Recombinant protein standard curve analysis (Anal. Biochem., 2024) |
| Detection Sensitivity | 1-10 pg/mL | 10-100 pg (total load) | Detection of low-abundance signaling phosphoproteins (Cell Rep. Methods, 2023) |
| Molecular Specificity | Specific for epitope | Specific for epitope + molecular weight | Analysis of protein isoforms in cell lysate (Sci. Rep., 2024) |
| Sample Volume Required | Low (50-100 µL) | Medium-High (20-50 µg total protein) | Mouse brain tissue homogenate study (Curr. Protoc., 2024) |
| Assay Time | 3-5 hours (hands-off) | 1-2 days (hands-on) | Standard protocol comparison (Nat. Protoc. Exchange, 2024) |
| Multiplexing Capability | Possible with panels | Limited (2-3 targets per blot) | Phospho-kinase profiling (Methods Mol. Biol., 2024) |
Detailed Experimental Protocols
Cited Experiment 1: Throughput & Precision Comparison (2023)
- Objective: Compare inter-assay variability for serum IL-6 measurement.
- ELISA Protocol: Commercial human IL-6 sandwich ELISA kit was used. 100 µL of serum (1:2 dilution) and standards were added to pre-coated wells. Following kit instructions, plates were incubated with detection antibody, streptavidin-HRP, and TMB substrate. Reaction stopped with H₂SO₄. Absorbance read at 450 nm with 570 nm correction.
- Western Blot Protocol: Serum proteins precipitated, resuspended in Laemmli buffer. Samples (equivalent to 5 µL serum/lane) separated on 12% Tris-Glycine SDS-PAGE, transferred to PVDF. Membrane blocked (5% BSA), probed with anti-IL-6 primary (1:1000, 4°C overnight) and HRP-conjugated secondary (1:5000, 1h). Detected via chemiluminescence, quantified by densitometry.
Cited Experiment 2: Sensitivity for Phospho-Protein Detection (2023)
- Objective: Determine lower detection limit for phosphorylated ERK1/2 in stimulated cell lysates.
- ELISA Protocol: Cell lysates were added to a plate pre-coated with an anti-total ERK capture antibody. Following incubation and wash, detection was performed sequentially with anti-phospho-ERK antibody, biotinylated secondary antibody, and streptavidin-HRP. Signal amplified via enzymatic amplification step.
- Western Blot Protocol: Lysates (25 µg total protein) separated on 10% Bis-Tris gel, transferred to nitrocellulose. Membranes were probed with anti-phospho-ERK1/2 (1:2000) and anti-total ERK (1:5000) antibodies simultaneously using different fluorescent dye-conjugated secondaries (800CW and 680LT). Imaging and quantification performed on a dual-channel infrared scanner.
Signaling Pathway Analysis Workflow
A typical workflow for studying a key pathway like MAPK/ERK using these techniques.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Primary Function in ELISA/WB | Key Consideration |
|---|---|---|
| High-Affinity, Validated Paired Antibodies | Capture & detection for sandwich ELISA; primary probe for WB. | Specificity validation (KO lysate) and matched pairs (ELISA) are critical. |
| HRP or AP Conjugates | Enzyme-linked detection for colorimetric/chemiluminescent signal. | Stability and minimal background activity differ between systems. |
| Chemiluminescent Substrate (e.g., ECL) | HRP substrate for WB detection; provides high sensitivity. | Choice between standard (fast) and high-sensitivity (prolonged) formulations. |
| TMB (3,3',5,5'-Tetramethylbenzidine) | Chromogenic HRP substrate for ELISA; generates measurable color change. | Stop solution (acid) required for reaction termination and plate reading. |
| Blocking Agent (e.g., BSA, Casein, Non-fat Milk) | Reduces non-specific binding to wells (ELISA) or membrane (WB). | Optimal agent depends on antibody and target; milk is unsuitable for phospho-studies. |
| Precision Microplate Reader | Measures absorbance in ELISA (450 nm). | Requires capability for dual-wavelength correction (e.g., 570 nm or 620 nm). |
| PVDF or Nitrocellulose Membrane | Solid support for immobilized proteins in WB. | PVDF offers higher protein binding and durability for re-probing. |
| Fluorescent Secondary Antibodies | Enable multiplex detection in WB via different emission wavelengths. | Requires a compatible imaging system (e.g., LI-COR Odyssey, Typhoon). |
Conclusion
ELISA and Western blot are complementary pillars of protein analysis, each with distinct strengths. ELISA excels in rapid, precise quantification of specific analytes in high-throughput or clinical settings, while Western blot provides critical information on protein size, modification state, and specificity in discovery research. The optimal choice is not a matter of which technique is universally superior, but which is most fit-for-purpose based on the experimental goals of quantification, characterization, throughput, and validation. Future directions point toward increased integration—using multiplex ELISA panels for broad screening followed by targeted Western blot confirmation, and leveraging advancements in digital Western blotting and ultrasensitive ELISA kits to push detection boundaries. For robust research and drug development, a strategic understanding of both methods is essential for generating reliable, reproducible, and clinically translatable data.